
Organic Chemistry
6th Edition
ISBN: 9781936221349
Author: Marc Loudon, Jim Parise
Publisher: W. H. Freeman
expand_more
expand_more
format_list_bulleted
Question
Chapter 28, Problem 28.41AP
Interpretation Introduction
Interpretation:
The reaction in Fig. P28.41 occurs as a sequence of two pericyclic reactions. The intermediate A, and the two reactions are to be stated.
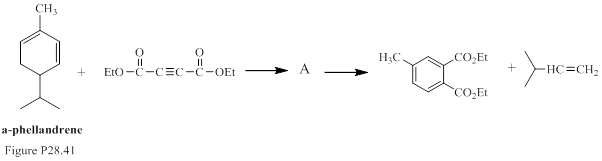
Concept introduction:
Commonly, cycloaddition reaction can be referred as a reaction of two different π-electron systems to form a ring product. Notably, in this reaction two new sigma bonds are formed in product with the disappearance of two fewer π-bonds. In other words, cyclo addition reaction can be best described as the reaction of number of electrons involved. In [4+2] cycloaddition one component is having four electrons and other component is having two electrons.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Which compound(s) is (are) best suited for nucleophilic aromatic substitution reactions?
Consider the tetracyclic compound with rings labeled A–D. (a) Which ring is the most reactive in electrophilic aromatic substitution? (b) Which ring is the least reactive in electrophilic aromatic substitution?
(a) Classify the enolates to 'kinetic' and 'thermodynamic.'
(b) Compare the stabilities of two enolates.
(c) Describe the three reaction conditions of 'thermodynamic enolates' and explain why.
Chapter 28 Solutions
Organic Chemistry
Ch. 28 - Prob. 28.1PCh. 28 - Prob. 28.2PCh. 28 - Prob. 28.3PCh. 28 - Prob. 28.4PCh. 28 - Prob. 28.5PCh. 28 - Prob. 28.6PCh. 28 - Prob. 28.7PCh. 28 - Prob. 28.8PCh. 28 - Prob. 28.9PCh. 28 - Prob. 28.10P
Ch. 28 - Prob. 28.11PCh. 28 - Prob. 28.12PCh. 28 - Prob. 28.13PCh. 28 - Prob. 28.14PCh. 28 - Prob. 28.15PCh. 28 - Prob. 28.16PCh. 28 - Prob. 28.17PCh. 28 - Prob. 28.18PCh. 28 - Prob. 28.19PCh. 28 - Prob. 28.20PCh. 28 - Prob. 28.21PCh. 28 - Prob. 28.22PCh. 28 - Prob. 28.23PCh. 28 - Prob. 28.24PCh. 28 - Prob. 28.25PCh. 28 - Prob. 28.26PCh. 28 - Prob. 28.27PCh. 28 - Prob. 28.28PCh. 28 - Prob. 28.29APCh. 28 - Prob. 28.30APCh. 28 - Prob. 28.31APCh. 28 - Prob. 28.32APCh. 28 - Prob. 28.33APCh. 28 - Prob. 28.34APCh. 28 - Prob. 28.35APCh. 28 - Prob. 28.36APCh. 28 - Prob. 28.37APCh. 28 - Prob. 28.38APCh. 28 - Prob. 28.39APCh. 28 - Prob. 28.40APCh. 28 - Prob. 28.41APCh. 28 - Prob. 28.42APCh. 28 - Prob. 28.43APCh. 28 - Prob. 28.44APCh. 28 - Prob. 28.45APCh. 28 - Prob. 28.46APCh. 28 - Prob. 28.47APCh. 28 - Prob. 28.48APCh. 28 - Prob. 28.49APCh. 28 - Prob. 28.50APCh. 28 - Prob. 28.51APCh. 28 - Prob. 28.52APCh. 28 - Prob. 28.53APCh. 28 - Prob. 28.54APCh. 28 - Prob. 28.55AP
Knowledge Booster
Similar questions
- (i) Explain why a high reaction temperature favours elimination reactions, instead of substitution reactions. (ii) Explain why polar aprotic solvents favour Sn2 reaction but not favour SN1 reaction.arrow_forwardthe following reaction scheme leads to the formation of compound C. give the structure of the final product C and of the intermediate products A and B and justify, using the mechanism, the formation of the product A. Give the serereochemistry of the final product obtainedarrow_forwardGive an example for each describe the following reactions (i) Hoffmann’s bromide reaction (ii) Gattermann reaction (in) A coupling reactionarrow_forward
- (b) Suggest a reasonable biosynthesis for the naturally occurring alkaloid coniine (isolated from hemlock), starting from octanoic acid. Coniinearrow_forwardSuggest reasonable mechanisms for each of the following reaction.arrow_forwardQuinapril (trade name Accupril) is used to treat high blood pressure and congestive heart failure. One step in the synthesis of quinapril involves reaction of the racemic alkyl bromide A with a single enantiomer of the amino ester B. (a) What two products are formed in this reaction? (b) Given the structure of quinapril, which one of these two products is needed to synthesize the drug?arrow_forward
- (a) Why are alkyl halides insoluble in water? (b) Why is Butan-l-ol optically inactive but Butan-2-ol is optically active? (c) Although chlorine is an electron withdrawing group, yet it is ortho-, Para- directing in electrophilic aromatic substitution reaction. Why?arrow_forwardDeduce the structure of compound C.arrow_forwardIdentify the pericyclic reactions in the following reaction schemes. Give the complete reaction name and indicate the course of the reaction with the aid of the arrow notation.arrow_forward
- (A) Provide the major organic product for the reaction below (B) Would the product be optically active of optically in active?arrow_forwardthe following reaction scheme leads to the formation of compound B. give the structure of the final products and of the intermediate product A and justify, using the mechanism, the formation of thesearrow_forwardQuinapril (trade name Accupril) is used to treat high blood pressure and congestive heart failure. One step in the synthesis of quinapril involves reaction of the racemic alkyl bromide A with a single enantiomer of the amino ester B. (a) What two products are formed in this reaction? (b) Given the structure of quinapril, which one of these two products is needed tosynthesize the drug?arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY