
Concept explainers
(a)
Interpretation:
The curved arrow mechanism for the reaction is to be stated and the reaction is to be classified as electrocyclic, cycloaddition, and the sigmatropic reaction. Along with that the number of electrons involved in the reaction is to be stated.
Concept introduction:
In the pericyclic reactions, the bond formation and dissociation occur simultaneously. The pericyclic reaction occurs in a concerted fashion with the cyclic shift of electrons. They are of three types, electrocyclic, cycloaddition and the sigmatropic reactions. They occur in the presence of heat or light.
Answer to Problem 28.1P
The given reaction is an example of an electrocyclic pericyclic reaction. The number of electrons involved in this reaction is six. The curved arrow mechanism is shown below.
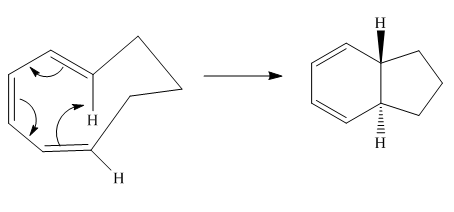
Explanation of Solution
The given reaction is an example of an electrocyclic reaction. This is because of in this electrocyclic reaction there is an intramolecular

Figure 1
The curved arrow mechanism for the reaction is shown in Figure 1. The reaction is a type of electrocyclic reaction. Along with it, the number of electrons involved in this reaction is six.
(b)
Interpretation:
The curved arrow mechanism for the reaction is to be stated and the reaction is to be classified as electrocyclic, cycloaddition, and the sigmatropic reaction. Along with that the number of electrons involved in the reaction is to be stated.
Concept introduction:
In the pericyclic reactions, the bond formation and dissociation occur simultaneously. The pericyclic reaction occurs in a concerted fashion with the cyclic shift of electrons. They are of three types electrocyclic, cycloaddition and the sigmatropic reactions. They occur in the presence of heat or light.
Answer to Problem 28.1P
The given reaction is an example of a sigmatropic pericyclic reaction. The number of electrons involved in this reaction is two. The curved arrow mechanism is shown below.
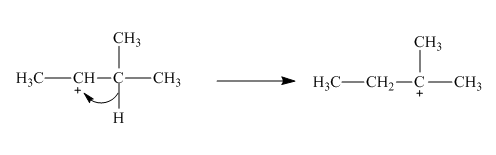
Explanation of Solution
The given reaction is an example of a sigmatropic reaction. This is because of in this sigmatropic reaction the allylic sigma bond present at one end of the system migrate to the other end of the system. The number of

Figure 2
The curved arrow mechanism for the reaction is shown in Figure 2. The reaction is a type of sigmatropic reaction. Along with it, the number of electrons involved in this reaction is two.
(c)
Interpretation:
The curved arrow mechanism for the reaction is to be stated and the reaction is to be classified as electrocyclic, cycloaddition, and the sigmatropic reaction. Along with that the number of electrons involved in the reaction is to be stated.
Concept introduction:
In the pericyclic reactions, the bond formation and dissociation occur simultaneously. The pericyclic reaction occurs in a concerted fashion with the cyclic shift of electrons. They are of three types electrocyclic, cycloaddition and the sigmatropic reactions. They occur in the presence of heat or light.
Answer to Problem 28.1P
The given reaction is an example of a cycloaddition pericyclic reaction. The number of electrons involved in this reaction is four. The curved arrow mechanism is shown below.
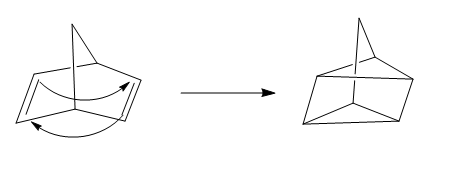
Explanation of Solution
The given reaction is an example of a cycloaddition reaction. This is because of in this cycloaddition reaction two

Figure 3
The curved arrow mechanism for the reaction is shown in Figure 3. The reaction is a type of cycloaddition reaction. Along with it, the number of electrons involved in this reaction is four.
(d)
Interpretation:
The curved arrow mechanism for the reaction is to be stated and the reaction is to be classified as electrocyclic, cycloaddition, and the sigmatropic reaction. Along with that the number of electrons involved in the reaction is to be stated.
Concept introduction:
In the pericyclic reactions, the bond formation and dissociation occur simultaneously. The pericyclic reaction occurs in a concerted fashion with the cyclic shift of electrons. They are of three types electrocyclic, cycloaddition and the sigmatropic reactions. They occur in the presence of heat or light.
Answer to Problem 28.1P
The given reaction is an example of a sigmatropic pericyclic reaction. The number of electrons involved in this reaction is six. The curved arrow mechanism is shown below.
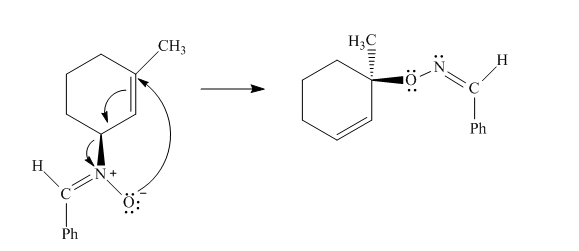
Explanation of Solution
The given reaction is an example of a sigmatropic reaction. This is because of in this sigmatropic reaction the allylic sigma bond present at one end of the

Figure 4
The curved arrow mechanism for the reaction is shown in Figure 4. The reaction is a type of sigmatropic reaction. Along with it, the number of electrons involved in this reaction is six.
(e)
Interpretation:
The curved arrow mechanism for the reaction is to be stated and the reaction is to be classified as electrocyclic, cycloaddition, and the sigmatropic reaction. Along with that the number of electrons involved in the reaction is to be stated.
Concept introduction:
In the pericyclic reactions, the bond formation and dissociation occur simultaneously. The pericyclic reaction occurs in a concerted fashion with the cyclic shift of electrons. They are of three types electrocyclic, cycloaddition and the sigmatropic reactions. They occur in the presence of heat or light.
Answer to Problem 28.1P
The given reaction is an example of a retro-electrocyclic pericyclic reaction. The number of electrons involved in this reaction is two. The curved arrow mechanism is shown below.
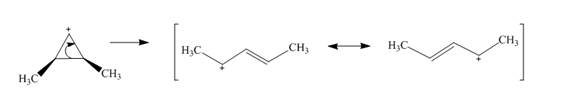
Explanation of Solution
The given reaction is an example of a retro-electrocyclic reaction. This is because of in the electrocyclic reaction there is an intramolecular

Figure 5
The curved arrow mechanism for the reaction is shown in Figure 5. The reaction is a type of retro-electrocyclic reaction. Along with it, the number of electrons involved in this reaction is two.
Want to see more full solutions like this?
Chapter 28 Solutions
Organic Chemistry
- (c) Compound 2F shown below is a tetra-substituted cyclooctatetraene. After heating 2F at 160 degree for 6 hours, researchers observed the formation of an isomer of 2F. Draw the isomer structure and provide a plausible mechanism to explain the formation of the isomers. 2Farrow_forwardIdentify the pericyclic reactions in the followingreaction schemes. Give the complete reaction name and indicate the course of the reaction with the aid of the arrow notation.arrow_forward(a) Explain how pyrrole is isoelectronic with the cyclopentadienyl anion.(b) Specifically, what is the difference between the cyclopentadienyl anion and pyrrole?(c) Draw resonance forms to show the charge distribution on the pyrrole structure.arrow_forward
- (a) When (Z)-3-methylhex-3-ene undergoes hydroboration–oxidation, two isomericproducts are formed. Give their structures, and label each asymmetric carbon atomas (R) or (S). What is the relationship between these isomers?(b) Repeat part (a) for (E)-3-methylhex-3-ene. What is the relationship between the productsformed from (Z)-3-methylhex-3-ene and those formed from (E)-3-methylhex-3-ene?arrow_forwardRewrite each of the following stereospecific transforms as a synthetic step in the forward direction, including reagents and any special reaction conditions. (a) (b) OH "CH3 CH3 H (Racemic) (c) OH (d) CN Br (Racemic) Brarrow_forward(a) H. H. 6.44 Which of the following reactions will yield a racemic mixture of products? HCI HBr (a) (c) H2 H2 (b) (d) Pt Ptarrow_forward
- Identify the pericyclic reactions in the followingreaction schemes. Give the complete reactionname and indicate the course of the reaction withthe aid of the arrow notation.arrow_forward(a) Name the reactant (including E/Z configuration) and draw the structure of the major product for the following reaction. + HBr (b) Explain your answer for the product in part (a), and include the structure of the carbocation intermediate in your explanation. (c) Draw the mechanism for the reaction in part (a), showing all intermediates and electron movement with arrows.arrow_forward5. Consider the following reaction: H-CI (ii) (i) intermediate product (a) Use curved arrows to indicate the electron flow in step (i). (b) Provide the line structure of the carbocation intermediate that forms in the reaction above. (c) Use curved arrows to show the electron flow in step (ii). (d) Explain briefly why the reaction in (ii) is regioselective.arrow_forward
- Using cyclohexane as your starting material, show how you would synthesize each of the following compounds. (Onceyou have shown how to synthesize a compound, you may use it as the starting material in any later parts of this problem.)(a) bromocyclohexane (b) cyclohexenearrow_forward(a) Draw the structures of the two possible products that can result from treating 1-ethylcyclohexene with hydrogen bromide and very briefly explain which one will be favoured). (b) Draw the curly arrow mechanism for the formation of the favoured productarrow_forwardAssign E or Z stereochemistry to each of the following alkenes, and convert each drawing into a skeletal structure (red = 0, yellow-green = Cl). (a) (b)arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





