
(a)
Interpretation:
A mechanism for each of the following transformations is to be suggested.
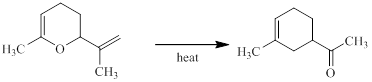
Concept introduction:
The Claisen rearrangement is a part of pericyclic reaction involving carbon-carbon bond formation. This reaction was discovered by Claisen and the reactions deals with the heating of allyl vinyl ether under thermal conditions to furnish an unsaturated carbonyl compound via [3, 3] sigmatropic rearrangement.
(b)
Interpretation:
A mechanism for each of the following transformations is to be suggested.

Concept introduction:
Electrocyclic reactions are a pericyclic reaction which occur intramolecularly. These reactions will result in the formation of ring compounds under the influence of heat or light. Notably, in this process one new sigma bond is formed and one old π-bond is consumed. Intriguingly, the reverse ring opening electrocyclic reaction can also be possible to occur under the same reaction mechanism but in reverse manner.
(c)
Interpretation:
A mechanism for each of the following transformations is to be suggested.
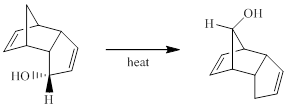
Concept introduction:
Generally, Sigmatropic reaction is referred as the migration of allylic sigma bond at one end of the π-electron system to the other end of the π-electron system as an uncatalyzed intramolecular reaction. Though, the position of π-bond is changed in Sigmatropic reaction, the total number of π-bonds remain unchanged. The sigma bond can be cleaved at the middle or at the end of the π-system. The formation of sigma bond at 3, 3-position of a 1, 5-diene is called as cope rearrangement.
Want to see the full answer?
Check out a sample textbook solution
Chapter 28 Solutions
Organic Chemistry
- A problem often encountered in the oxidation of primary alcohols to acids is that esters are sometimes produced as by-products. For example, oxidation of ethanol yields acetic acid and ethyl acetate: Propose a mechanism to account for the formation of ethyl acetate. Take into account the reversible reaction between aldehydes and alcohols:arrow_forwardHeterocyclic compounds can undergo nitration reactions with some regioselectivity, giving products in majority. From the following reaction give the raw material from which it is formed and the mechanism under which the transformation takes place.arrow_forwardConsider the following chemical transformation:The transformation takes place via twosequential pericyclic reactions. Identify the two reactions and give a critical explanationwhether the reactions are allowable.arrow_forward
- Devise concise syntheses for the following transformations. Clearly show the reagent andproduct for each step. All syntheses can be accomplished in two steps.arrow_forwardCompound A has molecular formula C7H15B.. Treatment of compound A with sodium ethoxide yields only one elimination product (compound B) and no substitution products. When compound B is treated with dilute sulfuric acid, compound C is obtained, which has molecular formula C7H160. Draw the structures of compounds A, B, and C.arrow_forwardShow how the following starting materials are converted to the given product by a series of two pericyclic reactions. Account for the observed stereochemistry.arrow_forward
- Account for any stereochemistry, major/minor products in the following reactions. Provide mechanistic explanations for your product(s).arrow_forwardShow how the following starting material is converted to the given product by a series of two pericyclic reactions. Account for the observed stereochemistry.arrow_forwardProvide IUPAC names for the following compounds. You must include stereochemical notations (E, Z, R, or S) where necessary.arrow_forward
- Show how you would synthesize the following compounds, starting with benzene or toluene and any necessary acyclic reagents. Assume para is the major product (and separable from ortho) in ortho, para mixtures.(a) 1-phenyl-1-bromobutane (b) 1-phenyl-1-methoxybutane (c) 3-phenylpropan-1-olarrow_forwardIdentify the reagents necessary to accomplish each of the following transformations.arrow_forwardConsider the tetracyclic aromatic compound drawn below, with rings labeled as A, B, C, and D. (a) Which of the four rings is most reactive in electrophilic aromatic substitution? (b) Which of the four rings is least reactive in electrophilic aromatic substitution? (c) What are the major product(s) formed when this compound is treated with one equivalent of Br2?arrow_forward
