
Concept explainers
(a)
Interpretation:
Each of the following compounds exists as a fluxional molecule that is interconverted into one or more identical forms by the sigmatropic process indicated. One structure in each case that demonstrates the process involved is to be drawn. Each process is an allowed pericyclic reaction – the statement is to be verified.
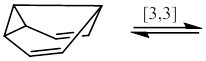
Concept introduction:
When a molecule undergoes a dynamic molecular motion involving the interchange of two or more groups which are chemically and magnetically different, then the molecule is said to have fluxionality nature. In this case, the signals corresponding to interchanged two or more groups will be identical, if the rate of exchange of the groups occurs very quickly. The fluxionality in organic molecules can be easily followed by multinuclear NMR spectroscopy.
(b)
Interpretation:
Each of the following compounds exists as a fluxional molecule that is interconverted into one or more identical forms by the sigmatropic process indicated. One structure in each case that demonstrates the process involved is to be drawn. Each process is an allowed pericyclic reaction – the statement is to be verified.
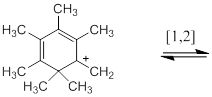
Concept introduction:
When a molecule undergoes a dynamic molecular motion involving the interchange of two or more groups which are chemically and magnetically different, then the molecule is said to have fluxionality nature. In this case, the signals corresponding to interchanged two or more groups will be identical, if the rate of exchange of the groups occurs very quickly. The fluxionality in organic molecules can be easily followed by multinuclear NMR spectroscopy.
Trending nowThis is a popular solution!

Chapter 28 Solutions
Organic Chemistry
- Consider the tetracyclic compound with rings labeled A–D. (a) Which ring is the most reactive in electrophilic aromatic substitution? (b) Which ring is the least reactive in electrophilic aromatic substitution?arrow_forward(i) Explain why a high reaction temperature favours elimination reactions, instead of substitution reactions. (ii) Explain why polar aprotic solvents favour Sn2 reaction but not favour SN1 reaction.arrow_forwardFor each of the following, write the major product(s) and then draw out each step in the mechanism using curved arrows. Show ALL lone pair electrons and formal charges. Redraw ALL molecules as to show explicitly ALL bonds being broken or formed. Identify the molecular orbital (HOMO) of the nucleophile and the molecular orbital (LUMO) of electrophile involved in the nucleophilic attack. MO diagrams are not necessary. (a). (b). -CHO 2. H₂O 1. LiAlH4 2. H₂O MgBr , etherarrow_forward
- (a) Discuss the aromaticity of a six membered heterocyclic compound which is weak base in nature. (b) Discuss about the stability factors of the reaction intermediates, which involved in a name reaction "Wittig rearrangement".arrow_forward(a) H. H. 6.44 Which of the following reactions will yield a racemic mixture of products? HCI HBr (a) (c) H2 H2 (b) (d) Pt Ptarrow_forwardThe natural product halomon could theoretically arise from another naturally occurring compound known as myrcene. To accomplish this, a biochemical process that could deliver the synthetic equivalent of BrCi to all three double bonds would be required. (Chem Comm. 2014, 50, 13725) (a) Using three molar equivalents of BrCL please provide a mechanism to account for the formation of the bracketed structure (you do not need to show stereochemistry in this mechanism) HB (3 equiv) myrcene balomon 8.61a Add curved arrow(s) to show the mechanism steps. Edit Drawing sitsarrow_forward
- Observe and identify what's the general reaction scheme and explain it.arrow_forwardGive IUPAC names for the following structures. (If appropriate, specify relative stereochemistry.) (a) (b) S Sarrow_forward(a) Give a mechanism for this reaction, showing how the two products arise as aconsequence of the resonance-stabilized intermediate.(b) The bromination of cyclohexene using NBS gives only one major product, as shown onthe previous page. Explain why there is no second product from an allylic shift.arrow_forward
- two positions of anthracene sometimes react more like polyenes thanlike aromatic compounds.(a) Draw a Kekulé structure that shows how the reactive positions of anthracene are the ends ofa diene, appropriate for a Diels–Alder reaction.(b) The Diels–Alder reaction of anthracene with maleic anhydride is a common organic labexperiment. Predict the product of this Diels–Alder reaction.arrow_forward(b) Discuss about the stability factors of the reaction intermediates, which involved in a name reaction "Wittig rearrangement".arrow_forward(a) suggest the suitable mechanism and illustrate the stepwise of the (heptyloxy)cyclopentane mechanism. (b) illustrate, and write the mechanism the formation of iodocyclopentane and 1-iodoheptane.arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





