
Interpretation:
Two amino acids have to be chosen and the dipeptide formed from the chosen amino acids has to be drawn. The formed dipeptide is whether polar, nonpolar or mixed has to be determined.
Concept introduction:
Amino acids are the molecules containing an
Carbon, hydrogen, oxygen and nitrogen are the key elements in amino acid.
General structure of an amino acid can be drawn as follows,
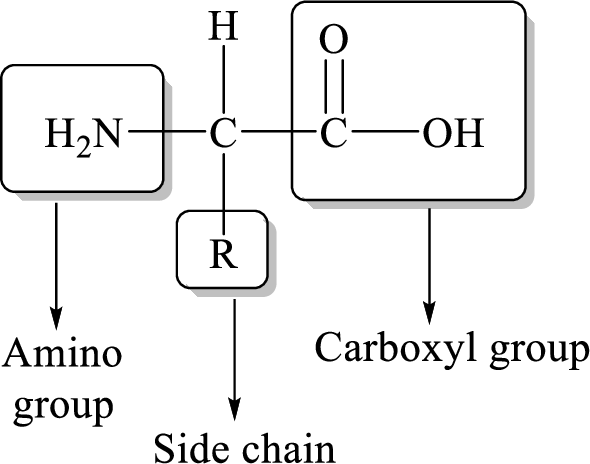
Peptide is a short
Peptide bonds are formed by a condensation reaction of carboxylic group of an amino acid and amino group of another amino acid with the removal of water molecule.
Want to see the full answer?
Check out a sample textbook solution
Chapter 12 Solutions
Chemistry In Context
- Practice question helparrow_forwardI need help solving thing and explanation on how you got this so I know what to do next timearrow_forwardComputation Mefenamic acid 500 mg Lactose 200 mg To make 1 cap Based on the given ingredients and amounts above, what capsule size should be used? Size 000 Size 1 Size 00 O Size 0arrow_forward
- Exercise 7) Make a molecular model of C2H6O. Now make another molecular model that is different from the first one (but don't take that one apart yet). Draw the structural formulas for both of your models in the space below, then write the condensed formula below each structure.arrow_forwardno need for explanation just answerarrow_forwardanswer quick please dont need explanationarrow_forward
- Which of characteristics below describes glycine? Check all that applyarrow_forwardExercise 6) Make a model ethylene, C2H4. Here you will need to use two of the longer, more flexible gray bonds for the double bond. Ask for help if you're not sure how to do this. Draw the structural formula and the write condensed formula below your structure.arrow_forwardreally needs help in Bio problem.arrow_forward
- Give a clear explanation detailed answerarrow_forwardWhy are soap molecules so attracted to calcium and magnesium ions? O calcium and magnesium ions have a charge of +2 O calcium and magnesium ions have a charge of 0 O calcium and magnesium ions have a charge of +1 O calcium and magnesium ions have a charge of -2 Submit Request Answer Next > Provide Feedbackarrow_forwardI need full details answers with accurate answer so, solve carefully. This is my only chance to get it right answer.arrow_forward
 Living By Chemistry: First Edition TextbookChemistryISBN:9781559539418Author:Angelica StacyPublisher:MAC HIGHER
Living By Chemistry: First Edition TextbookChemistryISBN:9781559539418Author:Angelica StacyPublisher:MAC HIGHER
