
Chemistry In Context
9th Edition
ISBN: 9781259638145
Author: Fahlman, Bradley D., Purvis-roberts, Kathleen, Kirk, John S., Bentley, Anne K., Daubenmire, Patrick L., ELLIS, Jamie P., Mury, Michael T., American Chemical Society
Publisher: Mcgraw-hill Education,
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 12, Problem 28Q
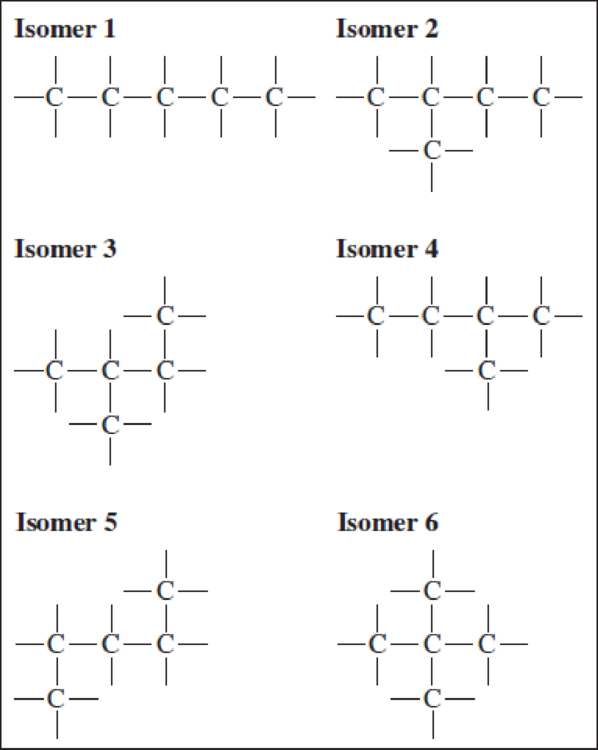
In Your Turn 12.12, you were asked to draw structural formulas for the three isomers of C5H12. One student submitted this set, with a note saying that six isomers had been found. Help this student see why some of the answers are incorrect.
Note: The hydrogen atoms have been omitted for clarity.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
1. CzH5OH(I) + HCOOH(I) → HCOOCzHs + H2O(I)
2. C₂H5OH(1)→ C₂H4(g) + H₂O(1)
3. C₂H4(9) + Cl₂(g) → C₂H4C12(1)
4. C6H6(1) + Br2(1)→ C6H5Br(1) + HBr(g)
5. 2C4H10(9) + 130₂(g) → 8CO₂(g) + 10H₂O(1)
6. 2C7H8(1) + 150₂(g) → 14CO(g) + 8H₂O(1)
Match each equation above with its reaction type.
Reaction Type
Addition
Complete combustion
Elimination
Esterification
Incomplete combustion
Substitution
Reaction Number from Above
→
◆
◆
Fill in the blanks to complete the following statement. One method used to differentiate between saturated and unsaturated compounds is to add a few drops of
orange-coloured aqueous bromine to samples of each organic compound. If the compound is
◆ the orange colour will quickly fade.
Determine the structures of the missing organic molecules in the following reaction:
Y
II
OH
X + H₂O
H+
H*
OH
Note: Molecules that share the same letter have the exact same structure.
In the drawing area below, draw the skeletal ("line") structures of the missing organic molecules X and Y. You may draw the structures in any arrangement
that you like, so long as they aren't touching.
Click and drag to start drawing a structure.
C
X
:0
Ś
m
c+
Draw structural formulas for the major organic product(s) of the reaction shown below.
OCH 3
FeCl3
+
Cl₂
•
You do not have to consider stereochemistry.
•
If no reaction occurs, draw the organic starting material.
•
•
Remember to include all of the formal charges on the atoms of any nitro groups.
Draw one structure per sketcher. Add additional sketchers using the drop-down menu in the bottom right corner.
•
Separate multiple products using the + sign from the drop-down menu.
?
ChemDoodleⓇ
Chapter 12 Solutions
Chemistry In Context
Ch. 12.1 - Skill Building Finding Equilibrium Glucose and...Ch. 12.1 - Prob. 12.3YTCh. 12.1 - Prob. 12.4YTCh. 12.2 - Prob. 12.5YTCh. 12.2 - Prob. 12.6YTCh. 12.2 - A practicing scientist must Judge a potential...Ch. 12.3 - Prob. 12.8YTCh. 12.3 - Skill Building Checking on Carbon a. Examine the...Ch. 12.3 - Prob. 12.10YTCh. 12.3 - Prob. 12.11YT
Ch. 12.3 - Prob. 12.12YTCh. 12.4 - Prob. 12.13YTCh. 12.4 - Skill Building Functional Groups in Dopamine Draw...Ch. 12.4 - Prob. 12.15YTCh. 12.5 - Prob. 12.16YTCh. 12.5 - Prob. 12.17YTCh. 12.6 - Prob. 12.18YTCh. 12.6 - Prob. 12.19YTCh. 12.6 - The structures of proteins, such as the ones shown...Ch. 12.7 - Reconsider your work in past chapters. List three...Ch. 12.7 - Prob. 12.22YTCh. 12.7 - Prob. 12.23YTCh. 12.8 - Prob. 12.24YTCh. 12.8 - Prob. 12.25YTCh. 12.9 - Skill Building Ester Formation Draw structural...Ch. 12.9 - Prob. 12.27YTCh. 12.9 - You Decide Supersize My Aspirin A friend who...Ch. 12.9 - Modern methods of drug discovery involve...Ch. 12.10 - Make two lists of drugs for each of the two...Ch. 12.10 - See for yourself the shapes of drug molecules by...Ch. 12.10 - Prob. 12.33YTCh. 12.10 - Prob. 12.34YTCh. 12 - Scientific Practices Follow the Hormone Using the...Ch. 12 - The field of chemistry has many sub-disciplines....Ch. 12 - Prob. 2QCh. 12 - Prob. 4QCh. 12 - Nitrous acid (HNO2) has a Ka value of 4.0 10 4,...Ch. 12 - Use the Henderson-Hasselbalch equation and Table...Ch. 12 - Write the structural formula and line-angle...Ch. 12 - Prob. 8QCh. 12 - Prob. 9QCh. 12 - Prob. 10QCh. 12 - Prob. 11QCh. 12 - Prob. 12QCh. 12 - Estradiol is relatively insoluble in water but...Ch. 12 - Prob. 14QCh. 12 - Prob. 15QCh. 12 - Prob. 16QCh. 12 - Define and relate the two terms: hormone and...Ch. 12 - Refer to Figure 11.17. Select two examples of...Ch. 12 - Prob. 19QCh. 12 - Molecules as diverse as cholesterol, sex hormones,...Ch. 12 - Prob. 21QCh. 12 - Prob. 22QCh. 12 - Prob. 23QCh. 12 - Sulfanilamide is the simplest sulfa drug, a type...Ch. 12 - Explain why an equilibrium constant cannot tell...Ch. 12 - Use the information in Table 12.1 to redraw Figure...Ch. 12 - Draw structural formulas for each of these...Ch. 12 - In Your Turn 12.12, you were asked to draw...Ch. 12 - Prob. 29QCh. 12 - Prob. 30QCh. 12 - Prob. 31QCh. 12 - Prob. 32QCh. 12 - Prob. 34QCh. 12 - Prob. 35QCh. 12 - Prob. 36QCh. 12 - Prob. 37QCh. 12 - Prob. 38QCh. 12 - The text states that some racemic mixtures contain...Ch. 12 - Prob. 40QCh. 12 - Prob. 41QCh. 12 - Prob. 44QCh. 12 - Prob. 47QCh. 12 - Prob. 49QCh. 12 - Dorothy Crowfoot Hodgkin first determined the...Ch. 12 - Prob. 52Q
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- In a lab, you are asked to determine the identity of 4 unknown compounds, A, B, C, and D. These compounds are ethane, ethanol, ethyl ethanoate, and ethanoic acid, but you don't know which is which. Using the table of physical properties below, identify each compound and justify your reasoning.arrow_forwardPlease help! I need to know how to draw the full chemical structure of isoproyl red. *I drew the structure of isopropyl alcohol (second pic) but was told this was incorrect and that the chemical structure needs to show a combination of methyl red and isopropyl alcohol.arrow_forward4. Decide whether each statement is true (T) or false (F). Place your answer in the blank space given. a. A hydrocarbon contains only atoms of carbon and hydrogen. b. Alkanes have the general formula C₂H₂- c. In a condensed structural diagram, the symbol "H" is used to show the position of a hydrogen atom. d. A continuous-chain alkane has one or more carbon branches attached to the parent chain. e. In a Lewis dot diagram of a hydrocarbon, each carbon-carbon and carbon-hydrogen bond is represented by a line segment. f. Any branch within branched alkanes is called an alkyl group. 1arrow_forward
- Draw structural formulas for organic products A and B in the window below. Br pentane Cul A B. • Draw only products having the organic portion of the original alkyl halide. • Draw carbon-lithium bonds using the single bond tool. If a structure has a copper-lithium bond, do not draw the lithium. Separate products from different steps using thesign from the drop-down menu.arrow_forward3a) The molecule ethyne (or acetylene), which has the molecular formula C₂H₂, contains two carbon atoms with linear geometry. Construct a model of ethyne by first connecting two black balls with three springs. Use two short sticks and two yellow balls to complete the structure. Sketch a perspective representation (a three dimensional drawing) of the structure.arrow_forward1, a)Write the sum of the coefficients of the balanced chemical equation for the complete combustion of acetylene (C2H2). Enter only a number for your answer and remember if the coefficient is implied to be 1, then you have to include that in your sum. b)Write the sum of the coefficients of the balanced chemical equation for the complete combustion of diethylether (C2H6O). Enter only a number for your answer and remember if the coefficient is implied to be 1, then you have to include that in your sum.arrow_forward
- Determine the structures of the missing organic molecules in the following reaction: H+ 뿜 XI + H₂O H+ Y Z Note: Molecules that share the same letter have the exact same structure. In the drawing area below, draw the skeletal ("line") structures of the missing organic molecules X, Y, and Z. You may draw the structures in any arrangement that you like, so long as they aren't touching. Molecule X shows up in multiple steps, but you only have to draw its structure once. Click and drag to start drawing a structure. X Śarrow_forwardPlease help me fill in the highlighted. I don't know. Please explain how to get the answers. Thank you in advance.arrow_forwardUsing family suffixes to name organic compounds Name these organic compounds: structure name H. H–C– OH H. CH, — СН,— СH, –C – H || Н— С — ОН Check Explanation O 2021 McGraw Hill LLC. AlI Rights Reserved. Tern ||arrow_forward
- Name this molecule. CH; CH, | CH,- CH, CH, CH, — С — сH,- сн — сн, — CH, | CH3 CH2arrow_forwardName 5 common organic compounds that are found in your home. Draw their Lewis structures and give their molecular formula. Encircle and identify the functional group/s that is/ are present in each molecule. Write your answers in another sheet of paper.arrow_forwarda student ran a TLC plate with three different molecules that have the same carbon structure but each contain a different functional groups. Help the student match the compounds to the correct lane of the TLC platearrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY
Acid-Base Titration | Acids, Bases & Alkalis | Chemistry | FuseSchool; Author: FuseSchool - Global Education;https://www.youtube.com/watch?v=yFqx6_Y6c2M;License: Standard YouTube License, CC-BY