
Concept explainers
An internally reversible refrigerator has a modified coefficient of performance accounting for realistic heat transfer processes of
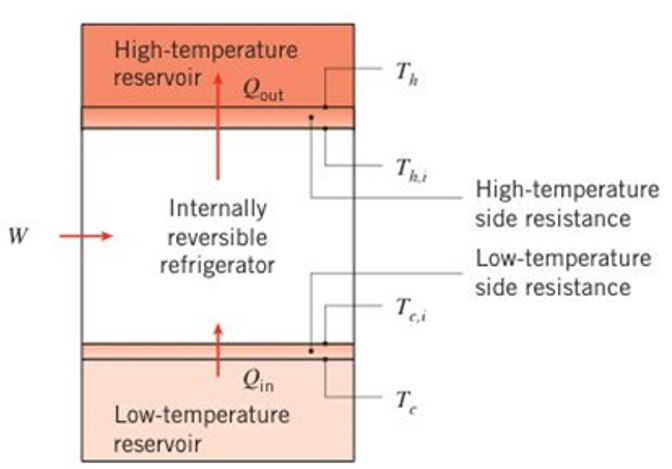
Want to see the full answer?
Check out a sample textbook solution
Chapter 1 Solutions
Introduction to Heat Transfer
- The operating condition for the single compressor in a household refrigerator is the lowest box temperature, which is typically A. 0F B. -20F C. 20F D. 40Farrow_forwardA new parabolic dish solar concentrator is being developed to supply electricity to a small remote community. The system operates with a Stirling engine connected to two heat reservoirs HR₁ and HR₂ as shown in Fig. Concentrated solar radiation Q˙ₛᵤₙ=200kW maintains HR₁ at constant temperature Tₕᵣ,₁=700℃, and a high performance heat sink device maintains HR₂ at temperature Tₕᵣ,₂=25℃. After some time of operation, an engineer working on the project realizes that the engine power output is performing below design specifications and only provides a power output of 120kW. After inspecting the system, they discover a contact resistance that causes the thermal communication between the two heat reservoirs and the engine to be imperfect in the sense that a finite temperature difference is required to establish heat transfer between each of the heat reservoirs and the engine. Sketch the cycle on a T-s diagram and include the temperatures of the heat reservoirs in your sketch. Calculate the…arrow_forwardChoose correct answer: Refrigerators and heat pumps operate on the same cycle but differ in their objectives. Select one: True Falsearrow_forward
- Between 900 K and T, (T, 400 K), another heat engine operates. For both to be equally efficient, the value of T, will bearrow_forwardReferring to the reversible heat pump cycle shown in the figure, p₁ = 14.7 lby/in². Pa = 34.7 lb/in², v₁ = 12.6 ft3/lb, v4 = 7.0 ft³/lb, and the gas is air obeying the ideal gas model. Step 1 Determine TH. in °R, and the coefficient of performance. Determine TH. in °R. TH= i Save for Later P4 °R PI V4 VI Ty Attempts: 0 of 4 used Submit Answer Activa Go to Searrow_forwardA heat engine with efficiency = 60 %, driving both a heat pump and refrigerator at the same time, the work generated by the heat engine (WH.E=W₁ + W₂). W₁ gose to heat pump which (COPH.P.= 2), and W₂ gose to Ref. From the Figure and missing 850 K Tmax=? Tmax =? values, Find: Pada =? Rada= 60 KJ a) The Qadd, Qrej, and Tmin of the Heat Pump, b) The Qadd, Qrej, and Tmax of the Heat Engine c) The Qrej, COPRef., and Tmax of the Refrigerator Qroj=? H.P. Tmin=? Qadd = ? W₁=150 KJ/ H.E. Qrej=? 75 K W₂=50 K/ Ref. Qrej=? 50 Karrow_forward
- 7. Find the refrigerating effect in a refrigerator if the COP is 3 and heat rejected is 40 kW.arrow_forwardReferring to the reversible heat pump cycle shown in the figure, p1 = 14.7 Ib/in?, p4 = 57.4 lbę/in?, v1 = 12.6 ft /lb, v4 = 5.0 ft /lb, and the gas is air obeying the ideal gas model. P p4 pi V4 V1 Determine TH, in °R, and the coefficient of performance.arrow_forwardA Carnot refrigerator operates in a room in which the temperature is (T„ = H 26°C). The refrigerator consumes 361W of power when operating. What is the temperature of the refrigerated space "T," (in 99 °C) when Q, = 7763W? %3D 7,arrow_forward
- Please derive the C.O.P of the refrigerator as a function of temperature using the temperature-entropy diagram. Assume ideal conditions. Thank you very mucharrow_forwardTrue or false Heat is transferred to a system by either a cheap commercially available air-sourced heat pump with coefficient of performance of just 1.5, or an expensive 100% efficient electrical resistance heater. The loss of Carnot Work Potential (Exergy destruction, or T0Sloss term) is minimum when heat is added by the cheap commercial heat pump. Heat is transferred to a system by either a cheap commercially available air-sourced heat pump with coefficient of performance of just 1.5, or an expensive 100% efficient isothermal heat source. The loss of Carnot Work Potential (Exergy destruction, or T0Sloss term) is minimum when heat is added by the isothermal heat source.arrow_forwardTwo reversible engines R1 and R2 are connected ins series between a heat source S and a cold body C. If T1 = 1200 R, T2 = 450 R, QH = 400 BTU and the engines have equal thermal efficiencies Determine the following a.) The temperature at which heat is rejected by R1 and received by R2 b.) The heat rejected to the cold bodyarrow_forward
 Refrigeration and Air Conditioning Technology (Mi...Mechanical EngineeringISBN:9781305578296Author:John Tomczyk, Eugene Silberstein, Bill Whitman, Bill JohnsonPublisher:Cengage Learning
Refrigeration and Air Conditioning Technology (Mi...Mechanical EngineeringISBN:9781305578296Author:John Tomczyk, Eugene Silberstein, Bill Whitman, Bill JohnsonPublisher:Cengage Learning
