
Organic Chemistry
12th Edition
ISBN: 9781118875766
Author: T. W. Graham Solomons, Craig B. Fryhle, Scott A. Snyder
Publisher: WILEY
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 9, Problem 10PP
PRACTICE PROBLEM 9.10
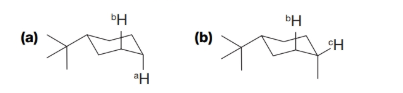
What is the dihedral angle and expected coupling constant between the labeled protons in each of the following molecules?
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
III) Can 1H NMR spectroscopy be used to differentiate between the two compounds? Briefly explain why or why not. Predict the 1H NMR spectrum for each compound (include integration, multiplicity, and approximate chemical shift). For each set, be sure that you put them in a data table format!
You are given two compounds, dimethylamine (CH3)2NH and ethanamide,
CH3CONH2.
a)
b)
c)
Which compound is more basic? Explain your answer.
How do you distinguish them by infrared spectroscopy?
Outline an extraction method to separate a mixture of these two compounds.
Among the five protons shown below select the most shielded (lowest chemical shift).
Hint: take into account the effects of an electronegative atom and likely resonance
structures.
H
H
HH
(1)
(2)
(3)
(4)
(5)
(1)
(2)
(3)
(4)
(5)
Chapter 9 Solutions
Organic Chemistry
Ch. 9 - Prob. 1PPCh. 9 - PRACTICE PROBLEM 9.2 What compound with molecular...Ch. 9 - PRACTICE PROBLEM 9.3
Using the method of Section...Ch. 9 - PRACTICE PROBLEM 9.4 How many signals would each...Ch. 9 - Prob. 5PPCh. 9 - Prob. 6PPCh. 9 - PRACTICE PROBLEM 9.7
The relative chemical shifts...Ch. 9 - Prob. 8PPCh. 9 - PRACTICE PROBLEM 9.9 Propose a structure for...Ch. 9 - PRACTICE PROBLEM 9.10
What is the dihedral angle...
Ch. 9 - PRACTICE PROBLEM 9.11 Draw the most stable chair...Ch. 9 - Prob. 12PPCh. 9 - PRACTICE PROBLEM 9.13 How many signals would you...Ch. 9 - Prob. 14PPCh. 9 - Prob. 15PPCh. 9 - Prob. 16PPCh. 9 - Prob. 17PPCh. 9 - PRACTICE PROBLEM 9.18
What are the expected ratios...Ch. 9 - Prob. 19PPCh. 9 - How many 1H NMR signals (not peaks) would you...Ch. 9 - How many 13C NMR signals would you predict for...Ch. 9 - Prob. 22PCh. 9 - Prob. 23PCh. 9 - Prob. 24PCh. 9 - Compound Q has the molecular formula C7H8. The...Ch. 9 - 9.26 Explain in detail how you would distinguish...Ch. 9 - Compound S (C8H16) reacts with one mole of bromine...Ch. 9 - A compound with molecular formula C4H8O has a...Ch. 9 - In the mass spectrum of 2, 6-dimethyl-4-heptanol...Ch. 9 - Prob. 30PCh. 9 - What are the masses and structures of the ions...Ch. 9 - Prob. 32PCh. 9 - Ethyl bromide and methoxybenzene (shown below)...Ch. 9 - 9.34 The homologous series of primary amines, ,...Ch. 9 - Propose a structure that is consistent with each...Ch. 9 - 9.36 Propose structures for compounds E and F....Ch. 9 - 9.37 Use the NMR and IR data below to propose a...Ch. 9 - 9.38 When dissolved in , a compound (K) with the...Ch. 9 - Compound T (C5H8O) has a strong IR absorption band...Ch. 9 - Prob. 40PCh. 9 - Deduce the structure of the compound that gives...Ch. 9 - Deduce the structure of the compound that gives...Ch. 9 - The 1H NMR spectrum of a solution of 1,...Ch. 9 - Acetic acid has a mass spectrum showing a...Ch. 9 - The 1H NMR peak for the hydroxyl proton of...Ch. 9 - The 1H NMR study of DMF (N, N-dimethylformamide)...Ch. 9 - 9.48 The mass spectra of many benzene derivatives...Ch. 9 - Prob. 49PCh. 9 - 1. Given the following information, elucidate the...Ch. 9 - Two compounds with the molecular formula C5H10O...
Additional Science Textbook Solutions
Find more solutions based on key concepts
Calculate the lattice energy of CaCl2 using a Born-Haber cycle and data from Appendices F and L and Table 7.5. ...
Chemistry & Chemical Reactivity
1. What did each of the following scientists contribute to our knowledge of the atom?
a. William Crookes
b. E...
Chemistry For Changing Times (14th Edition)
For Practice 1.1
Is each change physical or chemical? Which kind of property (chemical or physical) is demonst...
Principles of Chemistry: A Molecular Approach (3rd Edition)
Using the pKa values listed in Table 15.1, predict the products of the following reactions:
Organic Chemistry (8th Edition)
19. What is an ion?
Introductory Chemistry (6th Edition)
For each of the following molecules, (1) draw the correct Lewis structure; (2) label each polar covalent bond w...
General, Organic, and Biological Chemistry (3rd Edition)
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Please help answer the following question...thank you! Given the three potential constitutional isomers that you answered for problem 4, please examine the following 1H-NMR data, including integration and resonance assignments, and decide which of the molecules matches most closely with the spectral data. **I have attached the answer from problem 4 as well.arrow_forward6) Complete the spectroscopy data tables for a compound with molecular formula C6H12O. Determine the structure of the compound. Any labile protons, if they exist, will not be present in this particular 1H NMR spectrum.arrow_forward1) Complete the spectroscopy data tables for a compound with molecular formula C8H8O. Determine the structure of the compound. Any labile protons, if they exist, will not be present in this particular1H NMR spectrum.arrow_forward
- How would infrared spectroscopy be useful in distinguishing between the followingcompounds? Cite absorption bands you would expect from each.arrow_forwardThe 1HNMR signals the alkene protons of three ethylene derivatives with different substitution patterns are shown below. Considering that each of the derivatives contains AT MOST two substituents (X and/or Y) and at least two protons. A) Propose the possible substitution patterns for each of the derivatives based on the observed splitting patterns. Justify your proposals B) How might one differentiate between the three possible isomers of derivative 3 in the question above using 1H NMR spectroscopy? Explain in a sentence – no calculation is needed. C) Give TWO methods that can be used to differentiate between the three possible isomers of derivative 2 in the question above using 1H NMR spectroscopy? Explain in a sentence – no calculation is needed.arrow_forwardCan you explain spectroscopy? Let's start with NMR Spectroscopy. What is the difference between a proton being enantiotopic and homotopic? What does it mean for a proton to be shielded or deshielded? How do you determine J (the coupling constant)? What does 'coupling is mutual' mean? How is that translated on the NMS Spectroscopy spectrum? Does that have to do with the polarity of the molecule? Is there an easier way to explain that? When thinking about C-13 NRM Spectroscopy, what it meant by the statement, "the carbons cannot integrate"? When looking at an NMR Spectroscopy spectrum, how do you know which peak belongs to which proton? (The intensity of the peak and the ppm of the peak) P.S. These questions come from after watching a lecture. There is no graded assignment attached to these questions. I simply do not understand the material.arrow_forward
- Part 3B Set 2. Can 1H NMR spectroscopy be used to differentiate between the two compounds? Briefly explain why or why not. Predict the 1H NMR spectrum for each compound (include integration, multiplicity, and approximate chemical shift). Either draw the actual spectrum or put in a data table format.arrow_forwardPractice Problem 15.45 x Your answer is incorrect. Try again. How many signals do you expect in the ¹H NMR spectrum of the following compound. Isoprene A precursor for natural rubberarrow_forwardPredict the splitting pattern(s) you would expect for the proton(s) in the molecule below that will show in its NMR spectrum. Explain all answers.arrow_forward
- c) The two molecules below both possess a nitro group; however, one of the two nitro groups has a lower N=O stretching frequency its IR spectrum. Which molecule has the lower stretch (i.e. lower wavenumber value), and why? NO2 NO2 versusarrow_forward3. Predict the proton NMR spectrum of the following compound. Include number of peaks, chemical shifts, multiplicity and integrals in your answerarrow_forwardV.) A compound with molecular formula C8H10O has the following proton NMR spectrum. Determine the number of protons giving rise to each signal (a, b, c, and d). (SEE PICTURE ATTACHED).arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 EBK A SMALL SCALE APPROACH TO ORGANIC LChemistryISBN:9781305446021Author:LampmanPublisher:CENGAGE LEARNING - CONSIGNMENT
EBK A SMALL SCALE APPROACH TO ORGANIC LChemistryISBN:9781305446021Author:LampmanPublisher:CENGAGE LEARNING - CONSIGNMENT

EBK A SMALL SCALE APPROACH TO ORGANIC L
Chemistry
ISBN:9781305446021
Author:Lampman
Publisher:CENGAGE LEARNING - CONSIGNMENT
IR Spectroscopy; Author: Professor Dave Explains;https://www.youtube.com/watch?v=_TmevMf-Zgs;License: Standard YouTube License, CC-BY