
Concept explainers
Interpretation:The process that will occur measurably at room temperature should be chosen.
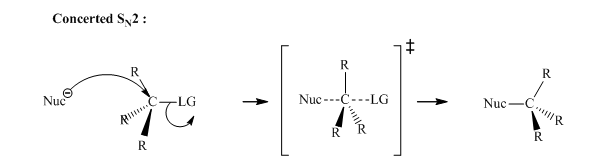
Concept introduction: Bimolecular substitution or proceeds via single-step mechanism. Thus it is well known as concerted mechanism. Nucleophile approaches carbon while the leaving group still departs from the rear side (opposite to leaving group). The transition state only illustrates the geometric orientation of the substrates and reagents as they pass through the maxima in the single-step me
A general
Polar-aprotic solvents accelerate the rate of
Want to see the full answer?
Check out a sample textbook solution
Chapter 6 Solutions
Organic Chemistry: Structure and Function
- Which mechanism(s) would be favored under the following reaction conditions?arrow_forwardPlease explain why this reaction will NOT WORK. NO2 1. HNO3, H2SO4, 2. EtCI, AICI3arrow_forwardFor a reversible reaction at an equilibrium: The rate of the reaction forward is the same as that of the reaction in reverse. The rate constant for the reaction forward is the same as that for the reverse. The free energy of substrate is the same as that of product. No conversion of substrate to product, and product to substrate occurs. Position of an equilibrium does not depend on substrate and product concentration , only on the magnitude of equilibrium constant.arrow_forward
- Match the reaction with the mechanism that it proceeds through. HO, HO. X catalytic H₂SO4 HBr Br₂ light 1. Free Radical Chain Reaction 2. SN1 3. SN2 4. E1 5. E2arrow_forwardWhich of the reactions has a product that is thermodynamically stable compared with the reactant?arrow_forwardCan you please help me draw all isomers of this compound and what is the relationships between these isomers, which isomers would react faster or slower?arrow_forward
- In each of the following three reaction coordinate diagrams, state: (a) Whether the reaction is exothermic or endothermic. (b) Whether the reaction is the slowest, the fastest, or intermediate in rate. (c) If all three reactions have the same entropy change between the reactant and product, which reaction has the largest favorable G0.arrow_forwardQ1. Draw the reaction mechanism using curly arrows to show each bond made/broken in the production of biodiesel from vegetable oil. Q2. NaOH is in a catalytic amount, why is it not needed in equimolar amount to the Methanol?arrow_forwardHow do you solve this mechanism?arrow_forward
- CHEMICAL REACTIONS: Illustrate and predict the products of the given chemical reactions below including the correct catalyst for each given reactions Hydration of Propanal.arrow_forward1) Sketch an energy diagram that would show the following:A) An exothermic reaction that is thermodynamically favorable and would occur quickly.B) An exothermic reaction that is thermodynamically favorable and would not occur at a measureablerate.C) An endothermic reaction that is thermodynamically favorable and would occur quickly.arrow_forwardConsider the following overall reaction, which will be discussed in Chapter 20.arrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning


