
a.
Interpretation: The product for the following reaction needs to be determined if reaction is possible:
Concept introduction: A chemical transformation of one substance (reactants) to the other (products) via single or several steps involved in the reaction is known as
b.
Interpretation: The product for the following reaction needs to be determined if reaction is possible:
Concept introduction: A chemical transformation of one substance (reactants) to the other (products) via single or several steps involved in the reaction is known as chemical reaction.
c.
Interpretation: The product for the following reaction needs to be determined if reaction is possible:
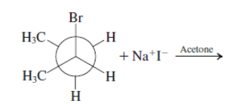
Concept introduction: A chemical transformation of one substance (reactants) to the other (products) via single or several steps involved in the reaction is known as chemical reaction.
d.
Interpretation: The product for the following reaction needs to be determined if reaction is possible:
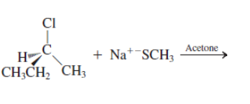
Concept introduction: A chemical transformation of one substance (reactants) to the other (products) via single or several steps involved in the reaction is known as chemical reaction.
e.
Interpretation: The product for the following reaction needs to be determined if reaction is possible:
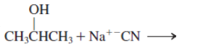
Concept introduction: A chemical transformation of one substance (reactants) to the other (products) via single or several steps involved in the reaction is known as chemical reaction.
f.
Interpretation: The product for the following reaction needs to be determined if reaction is possible:
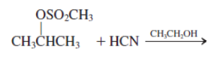
Concept introduction: A chemical transformation of one substance (reactants) to the other (products) via single or several steps involved in the reaction is known as chemical reaction.
g.
Interpretation: The product for the following reaction needs to be determined if reaction is possible:
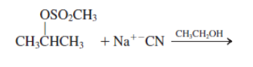
Concept introduction: A chemical transformation of one substance (reactants) to the other (products) via single or several steps involved in the reaction is known as chemical reaction.
h.
Interpretation: The product for the following reaction needs to be determined if reaction is possible:
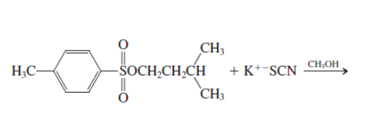
Concept introduction: A chemical transformation of one substance (reactants) to the other (products) via single or several steps involved in the reaction is known as chemical reaction.
i.
Interpretation: The product for the following reaction needs to be determined if reaction is possible:
Concept introduction: A chemical transformation of one substance (reactants) to the other (products) via single or several steps involved in the reaction is known as chemical reaction.
j.
Interpretation: The product for the following reaction needs to be determined if reaction is possible:
Concept introduction: A chemical transformation of one substance (reactants) to the other (products) via single or several steps involved in the reaction is known as chemical reaction.
Want to see the full answer?
Check out a sample textbook solution
Chapter 6 Solutions
Organic Chemistry: Structure and Function
- Write the IUPAC name of the product of this reaction: HBr CH2=C(CH3)2arrow_forwardPredict the products of the following reaction. Be sure your chemical equation is balanced! (CH)() + O2(g) →arrow_forwardPredict the products of the following reaction. If no reaction will occur, use the NO REACTION button. Be sure your chemical equation is balanced! CH3(CH)2(CH2)2 CH3(l) + O2(g) ->arrow_forward
- Consider the following chemical reaction. HNO3, H2SO4 Predict the product of this reaction. Select the name that best corresponds to the product. O 2,4,6-trinitrotoluene O sulfuric acid O benzene O nitrobenzenearrow_forwardWhat are the products of the complete combustion of 1-propanol, C,H,OH? A B C D carbon and oxygen carbon dioxide and water carbon monoxide and water carbon and hydrogenarrow_forwardWhat is the product (name and stable structure) formed from the reaction:CH3CH2C≡CCH2CH3 + H2O (H2SO4 as catalyst) →arrow_forward
- Suggest a simple chemical test that will differentiate between the following pairs of compounds. Write equations for the reactions involved. ▪ phenol and isopentyl alcohol ▪ tert-butyl alcohol and isobutyl alcohol ▪ neopentyl alcohol and ether ▪ sec-butyl alcohol and neopentyl alcohol ▪ propene and 2-butanolarrow_forwardDraw structural formulas to complete and balance the following reactions. butanoic acid + ethanol → propanoic acid + methanol → butanoic acid + 1-butanol → ethanoic acid + methanol → pentanoic acid + 2-butanol → hexanoic acid + 2-propanol → methanoic acid + 3-heptanol → propanoic acid + 1-propanol → ethanoic acid + 2-pentanol → octanoic acid + ethanol →arrow_forwardWhat is the complete combustion reaction for 2,2-dimethylpentane?arrow_forward
- Complete the following reaction by writing the name of the product. ch3coh + H2 ?arrow_forwardDraw the organic products of the chlorination of propane in a limited supply of chlorine gas. Cl Cl H H HH H Н—с—с—с—н +с, — H H H Incorrectarrow_forwardb) Ho わ-fャコーfつ Koc 5. Complete the following oxidation reactions for alcohols. Draw the structure of the product. Name the reactant and identify the type of compound formed in the product. CHっーCHューC OH b) CHy CH3arrow_forward
 Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning


