
Organic Chemistry (9th Edition)
9th Edition
ISBN: 9780321971371
Author: Leroy G. Wade, Jan W. Simek
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 12, Problem 12.29SP
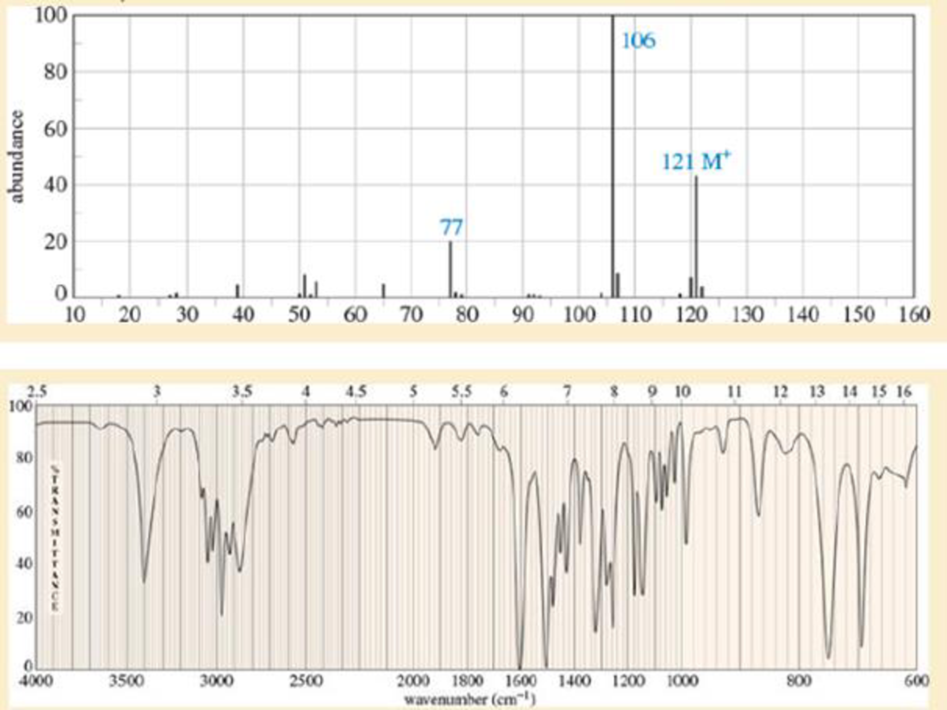
The ultimate test of fluency in MS and IR is whether you can determine a moderately complex structure from just the MS and the IR, with no additional information. The IR and MS of a compound are shown below. Use everything you know about IR and MS, plus reasoning and intuition, to determine a likely structure. Then show how your proposed structure is consistent with these spectra.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Determine the structure for the molecule that most likely produced these spectra. Use all pictures
Counterfeit drugs are a common problem in developing regions of the world. Oftentimes, counterfeit pills are made with compounds such as lactose. A lab technician has obtained the IR spectrum shown above for a sample reported to be citalopram, an antidepressant drug. Does the IR spectrum belong to citalopram or lactose? Explain your answer by describing what feature of the IR spectrum confirms your choice and describe what feature is missing from the IR spectrum for the other compound.
A. citalopram
B. lactose
8. A strong signal in infrared spectroscopy indicates that a molecule matches the emitted
electromagnetic radiation and reports a high transmittance.
True
False
9. The signals observed from the C-C bond in an alkene will report at a higher wavenumber than
the C-C bond in an alkyne.
True
False
10. The electronegativity difference present in a dipole moment within a bond is directly
proportional to the electromagnetic field produced.
True
False
Chapter 12 Solutions
Organic Chemistry (9th Edition)
Ch. 12.3 - Complete the following conversion table. (cm1)...Ch. 12.5 - Which of the bonds shown in red are expected to...Ch. 12.7C - For each hydrocarbon spectrum, determine whether...Ch. 12.9A - Spectra are given for three compounds. Each...Ch. 12.10 - The infrared spectra for three compounds are...Ch. 12.12 - Prob. 12.6PCh. 12.14B - Identify which of these four mass spectra indicate...Ch. 12.15A - Show the fragmentation that accounts for the...Ch. 12.15A - Show the fragmentations that give rise to the...Ch. 12.15B - Ethers are not easily differentiated by their...
Ch. 12.15C - Prob. 12.11PCh. 12 - Prob. 12.12SPCh. 12 - Prob. 12.13SPCh. 12 - All of the following compounds absorb infrared...Ch. 12 - Prob. 12.15SPCh. 12 - Four infrared spectra are shown, corresponding to...Ch. 12 - Predict the masses and the structures of the most...Ch. 12 - Prob. 12.18SPCh. 12 - Prob. 12.19SPCh. 12 - (A true story) While organizing the undergraduate...Ch. 12 - Prob. 12.21SPCh. 12 - Prob. 12.22SPCh. 12 - An unknown, foul-smelling hydrocarbon gives the...Ch. 12 - covered a synthesis of alkynes by a double...Ch. 12 - Three IR spectra are shown, corresponding to three...Ch. 12 - Prob. 12.26SPCh. 12 - Prob. 12.27SPCh. 12 - Prob. 12.28SPCh. 12 - The ultimate test of fluency in MS and IR is...Ch. 12 - Prob. 12.30SPCh. 12 - Consider the following four structures, followed...
Additional Science Textbook Solutions
Find more solutions based on key concepts
Draw a Lewis structure for each covalent molecule. a. HBr b. CH3F c. H2O2 d. N2H4 e. C2H6 f. CH2Cl2
Principles of General, Organic, Biological Chemistry
Write the electron configurations far each of the following elements: (a) Sc. (b) Ti. (c) Cr. (d) Fe. (e) Ru
Chemistry by OpenStax (2015-05-04)
4. 38 Strontium has four naturally occurring isotopes, with mass numbers 84, 86, 87, arid 88.
a. Write the atom...
General, Organic, and Biological Chemistry: Structures of Life (5th Edition)
Practice Exercise 1
Which of the following factors determines the size of an atom? a. the volume of the nucleus...
Chemistry: The Central Science (14th Edition)
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Infrared spectroscopy is most useful for Please choose the correct answer from the following choices, and then select the submit answer button. Answer choices determining the molecular structure of an unknown compound. differentiating between functional groups. determining the exact chemical formula of an unknown compound. determining the molecular mass.arrow_forwardi need help with interpreting these spectra to determine molecular structure, all 3 spectra correlate to the same molecule.arrow_forwardFrom the MS data shown below, identify the unknown organic molecule. Indicate any partial structures that you are able to deduce from these spectra and explain how you identify these.arrow_forward
- Explain why this is the correct spectra for these molecules. Identify a specific absorption band which identifies each characteristic functional group of the chosen molecule.arrow_forwardYour classmate needs to decide if the spectrum shown below is a 13CNMR or an 1HNMR spectrum of 2-methyl propane. What is your advice to the student and why?arrow_forwardThis is an infrared spectra of a compound whose molar mass is 108 g/mol. Determine the molecular formula and propose a structure.arrow_forward
- Can you break down this Infrared Spectroscopy? Break down the different peaks and what they could bearrow_forward1. how aas work? 2. how uv-vis work? 3. how nmr work? 4. how infrared spectroscopy work? 5. how molecular spectroscopy work?arrow_forward4. For each example below, draw at least onc possīble isomer that is consistent with the molecular formula and the associated IR spectrum. Page 3 a. MW 82, CaH1O be 48 20 1000 b. MW 116, Coll2O2 4000 2540 SL LOB2 9262 2042arrow_forward
- how molecular spectroscopy work?arrow_forwardThe IR spectra of two isomers are given below. Identify which IR spectra belong to isopropylbenzene and propylbenzene, respectively. How we decide them?arrow_forward6. Provide two possible Lewis structures for the formula: C,H,O (For help, you may want to review the material in the next lab or Chapter 12, section 4 in your textbook). Dbe ress ic the h d peccop ecomputer-bas appear to be Remember that signals in IR spectroscopy are due to bonds vibrating. What similar and different bonds found in the two structures you drew in question 6? Would you expect the spectra to look the same or different? Explain. 7. hearrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning
Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:9781305079113
Author:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning
Molecular spectroscopy; Author: Vidya-mitra;https://www.youtube.com/watch?v=G6HjLIWvCQo;License: Standard YouTube License, CC-BY