
Concept explainers
a)
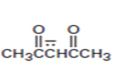
Interpretation:
The resonance structures for the anion shown are to be drawn.
Concept introduction:
Resonance forms differ only in the position of the π bonding and nonbonding pair of electrons. The position and hybridization of the atoms do not change from one resonance form to the other. Octet rule is applied while writing the different resonance forms.
b)
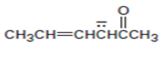
Interpretation:
The resonance structures for the anion shown are to be drawn.
Concept introduction:
Resonance forms differ only in the position of the π bonding and nonbonding pair of electrons. The position and hybridization of the atoms do not change from one resonance form to the other. Octet rule is applied while writing the different resonance forms.
c)
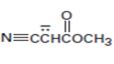
Interpretation:
The resonance structures for the anion shown are to be drawn.
Concept introduction:
Resonance forms differ only in the position of the π bonding and nonbonding pair of electrons. The position and hybridization of the atoms do not change from one resonance form to the other. Octet rule is applied while writing the different resonance forms.
d)
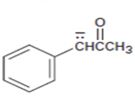
Interpretation:
The resonance structures for the anion shown are to be drawn.
Concept introduction:
Resonance forms differ only in the position of the π bonding and nonbonding pair of electrons. The position and hybridization of the atoms do not change from one resonance form to the other. Octet rule is applied while writing the different resonance forms.
e)
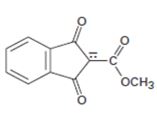
Interpretation:
The resonance structures for the anion shown are to be drawn.
Concept introduction:
Resonance forms differ only in the position of the π bonding and nonbonding pair of electrons. The position and hybridization of the atoms do not change from one resonance form to the other. Octet rule is applied while writing the different resonance forms.
Trending nowThis is a popular solution!

Chapter 22 Solutions
Organic Chemistry
- 51. Which is NOT a valid resonance structure for the conjugate base of this molecule? (A) (C) S (A) OEt O :O: : OEt HBr ROOR OCH 3 (B) 52. What is the major product of this reaction sequence? O PPh3 (D) 11 Mg ether (B) de OEt :0 aom Сосно LOCH 3 53. Which reagent is necessary for this transformation? (D) H OEt H OEt CH31 PPh3 ACS 56.arrow_forwardWhich of the following are correct resonance structures for benzophenone shown below? کر ک ن Benzophenone A В C D O A and Barrow_forwardCompound P has molecular formula C5H9ClO2. Deduce the structure of P from its 1H and 13C NMR spectra.arrow_forward
- Compound X (molecular formula C10H12O) was treated with NH2NH2,−OH to yield compound Y (molecular formula C10H14). Based on the 1H NMR spectra of X and Y given below, what are the structures of X and Y?arrow_forwardElectrophilic aromatic substitution usually occurs at the 1-position of naphthalene, also called the a position. Predict themajor products of the reactions of naphthalene with the following reagents. cyclohexanol and BF3arrow_forwardFor following substituted benzenes: [1] C6H5Br; [2] C6H5CN; [3] C6H5OCOCH3: Does the substituent activate or deactivate the benzene ring inelectrophilic aromatic substitution?arrow_forward
- solvent such as hydrocarbon, neither accept nor donate protons.arrow_forwardThe compound MON-0585 is a non-toxic, biodegradable larvicide that is highly selective against mosquito larvae. Synthesize MON-0585 using either benzene or phenol as a source of the aromatic rings.arrow_forwardProntosil, an azo dye, is a prodrug. It is metabolized in the body to the sulfa drug sulfanilamide, the active antibiotic. Show the arenediazonium ion and the aromatic compound that form the azo compound Protosil. S-NH2 H2N- N- NH2arrow_forward
- The 1H and 13C NMR spectra below belong to a compound with formula C6H10O2. Propose a structure for this compound.arrow_forward1, 6-Methanonaphthalene has an interesting 1H NMR spectrum in which the eight hydrogens around the perimeter absorb at 6.9 to 7.3 δ, while the two CH2 protons absorb at -0.5 δ. Tell whether it is aromatic, and explain its NMR spectrum.arrow_forwardHow would you explain the o-p directing formation of phenol, while nitro benzene is m directional? Resonance. Describe by showing their structures.arrow_forward

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

