
a)
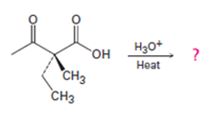
Interpretation:
Whether the
Concept introduction:
The decarboxylation of a β- keto acid takes place through the formation of a planar enol intermediate which then tautomerizes to the keto form. If the β- keto acid is optically active and if the chiral centre is involved in the reaction, the ketone product will be racemic and optically inactive. If the chiral centre is not involved in the reaction then the optical activity is retained in the ketone product.
To state:
Whether the ketone obtained by the decarboxylation of the optically active β- keto acid will be optically active or not.
To propose:
A mechanism to explain the formation of the ketone.
b)
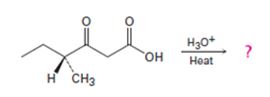
Interpretation:
Whether the ketone obtained by the decarboxylation of the optically active β- keto acid will be optically active or not is to be stated. A mechanism to explain the formation of the ketone is to be proposed.
Concept introduction:
The decarboxylation of a β- keto acid takes place through the formation of a planar enol intermediate which then tautomerizes to the keto form. If the β- keto acid is optically active and if the chiral centre is involved in the reaction, the ketone product will be racemic and optically inactive. If the chiral centre is not involved in the reaction then the optical activity is retained in the ketone product.
To state:
Whether the ketone obtained by the decarboxylation of the optically active β- keto acid will be optically active or not.
To propose:
A mechanism to explain the formation of the ketone.
Trending nowThis is a popular solution!

Chapter 22 Solutions
Organic Chemistry
- The following molecule undergoes an intramolecular reaction in the presence of pyrrolidinium acetate, the protonated form of pyrrolidine. Draw the product of this reaction, assuming that a dehydration reaction takes place.arrow_forwardBased on the image attached, it shows compound X. Suggests the synthesis pathways (not more than two reactions) for the formation of any ketone compound by using Compound X as a precursor. Use any reactions that are relatable.arrow_forwardWrite the mechanism for the following reactions: 1. the acid-catalyzed hydrolysis of an imine to a carbonyl compound and a primary amine 2. the acid-catalyzed hydrolysis of an enamine to a carbonyl compound and a secondary amine a. How do the two mechanisms differ?arrow_forward
- The most stable conformation of most aldopyranoses is one in which the largest group, the CH2OH group, is equatorial. However, alpha-D-idopyranose exists primarily in a conformation with an axial CH2OH group. Write formulas for the two chair conformations of a-D-idopyranose (one with the CH2OH group axial and one with the CH2OH group equatorial) and provide an explanationarrow_forwardShow how acid derivatives hydrolyze to carboxylic acids under either acidic or basicconditions. Explain why some acid derivatives (amides, for example) require muchstronger conditions for hydrolysis than other derivatives.arrow_forwardIn an attempt to synthesize compound C through a two-step process, a chemist discovered after completing the first step that they had inadvertently produced two distinct compounds, A and B. Upon examining the infrared spectroscopy (IR) results, it was observed that both A and B exhibited peaks indicative of a ketone and an ester group. Please provide the molecular structures of A and B. OEt NaOEt ΕΙΟ A B In a chemical experiment, they noticed that both components, A and B, from a combined sample turned into a new compound, C, during the following stage. The task is to determine what compound C looks like and explain how compound A or B changes into compound C through a reaction. Compound C should be the primary molecule containing carbon created in this process, not just a by-product. A B H3O+, H₂O, A Mechanism = сarrow_forward
- The ketone shown was prepared in a three-step sequence from ethyl trifluoroacetate. The first step in the sequence involved treating ethyl trifluoroacetate with ammonia to give compound A. Compound A was in turn converted to the desired ketone by way of compound B. Fill in the missing reagents in the sequence shown, and give the structures of compounds A and B.arrow_forwardOne of the key components in fatty acid synthesis is acetoacetyl‑ACP, which is formed from a Claisen condensation of a fatty acid thioester (acetyl synthase, simplified here) with the enolate formed from the decarboxylation of malonyl ACP. The given steps show the first round of this elongation. Draw a curved arrow mechanism to show how these reactions give acetoacetyl‑ACP.arrow_forwardProvide a synthesis pathway to get the productsarrow_forward
- What is the hydrolysis in the following acetal in aqueous acidarrow_forwardThe Stork reaction is a condensation reaction between an enamine donor and an α,β-unsaturated carbonyl acceptor. The overall reaction consists of a three-step sequence of formation of an enamine from a ketone, Michael addition to an α,β-unsaturated carbonyl compound, and hydrolysis of the enamine in dilute acid to regenerate the ketone. Consider the Stork reaction between acetophenone and 3-buten-2-one. Draw the structure of the product of the enamine formed between acetophenone and pyrrolidine. Draw the structure of the Michael addition product. Draw the structure of the final product.arrow_forwardShow how the following ketones might be synthesized from the indicated acids, usingany necessary reagents.(b) methyl cyclohexyl ketone from cyclohexanecarboxylic acidarrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning


