
Organic Chemistry (9th Edition)
9th Edition
ISBN: 9780321971371
Author: Leroy G. Wade, Jan W. Simek
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 21.6, Problem 21.14P
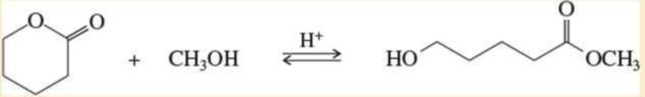
Propose a mechanism for the following ring-opening transesterification. Use the mechanism in Problem21-13 as a model
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Ch19-5: Which of the compounds is the intermediate called hemiacetal of the
following reaction?
요
HO OH H₂SO
(-H₂O)
OH
HO
OH
HO
HOO
O
OH
OH
The rate constant for the uncatalyzed reaction of two molecules of glycine ethyl ester to form glycylglycine ethyl ester is 0.6 M-1s-1. In the presence of Co2+, the rate constant is 1.5 x 106 M-1s-1. What rate enhancement does the catalyst provide?
Amantadine is effective in preventing infections caused by the influenza A virus and in
treating established illnesses. It is thought to block a late stage in the assembly of the
virus. Amantadine is synthesized by treating 1-bromoadamantane with acetonitrile in
sulfuric acid to give N-adamantylacetamide, which is then converted to amantadine.
CH,C=N
in H,SO,
Br
NHCCH3
NH2
1-Bromoadamantane
Amantadine
(a) Propose a mechanism for the transformation in Step 1.
(b) Describe experimental conditions to bring about Step 2.
Chapter 21 Solutions
Organic Chemistry (9th Edition)
Ch. 21.2F - Name the following carboxylic acid derivatives,...Ch. 21.4A - Prob. 21.2PCh. 21.4A - Prob. 21.3PCh. 21.4A - Prob. 21.4PCh. 21.5C - Prob. 21.7PCh. 21.6 - When ethyl 4-hydroxybutyrate is heated in the...Ch. 21.6 - Propose a mechanism for the following ring-opening...Ch. 21.6 - Prob. 21.15PCh. 21.7B - Prob. 21.16PCh. 21.7C - Prob. 21.19P
Ch. 21.7C - Prob. 21.20PCh. 21.7C - Prob. 21.21PCh. 21.7D - Prob. 21.22PCh. 21.7D - The mechanism for acidic hydrolysis of a nitrile...Ch. 21.8A - Prob. 21.24PCh. 21.8C - Prob. 21.25PCh. 21.9 - Prob. 21.26PCh. 21.9 - Prob. 21.27PCh. 21.9 - Prob. 21.28PCh. 21.10 - Draw a mechanism for the acylation of anisole by...Ch. 21.10 - Prob. 21.30PCh. 21.11 - Prob. 21.31PCh. 21.11 - Prob. 21.32PCh. 21.12 - Problem 21-33 Propose a mechanism for the...Ch. 21.12 - Suggest the most appropriate reagent for each...Ch. 21.12 - Show how you would synthesize each compound,...Ch. 21.13 - Prob. 21.36PCh. 21.13 - Prob. 21.37PCh. 21.14 - Prob. 21.38PCh. 21.14 - Prob. 21.39PCh. 21.16 - Prob. 21.40PCh. 21.16 - Prob. 21.41PCh. 21 - Prob. 21.42SPCh. 21 - Give appropriate names for the following...Ch. 21 - Predict the major products formed when benzoyl...Ch. 21 - Predict the products of the following reactions....Ch. 21 - Prob. 21.46SPCh. 21 - Prob. 21.47SPCh. 21 - Prob. 21.48SPCh. 21 - Propose mechanisms for the following reactions.Ch. 21 - Prob. 21.51SPCh. 21 - An ether extraction of nutmeg gives large...Ch. 21 - Prob. 21.53SPCh. 21 - Show how you would accomplish the following...Ch. 21 - Prob. 21.55SPCh. 21 - Prob. 21.56SPCh. 21 - Prob. 21.57SPCh. 21 - Prob. 21.58SPCh. 21 - Prob. 21.59SPCh. 21 - Explain this curious result. What does this...Ch. 21 - Prob. 21.61SPCh. 21 - Prob. 21.62SPCh. 21 - Prob. 21.63SPCh. 21 - A chemist was called to an abandoned aspirin...Ch. 21 - Prob. 21.67SPCh. 21 - The IR spectrum, 13ONTVTR spectrum, and 1HNMR...Ch. 21 - Prob. 21.69SPCh. 21 - Prob. 21.70SPCh. 21 - Prob. 21.71SP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Naturally occurring compounds called terpenoids, which we'll discuss in Section 27-5, are biosynthesized by a pathway that involves loss of CO2 from 3-phosphomevalonate 5-diphosphate to yield isopentenyl diphosphate. Use curved arrows to show the mechanism of this reaction.arrow_forwardProvide the mechanism for the following transformation: NaCN CN + Naciarrow_forwardProvide the product(s) for the following reaction: H₂N₂Barrow_forward
- 13-29 Show that if you add Steps 2a and 2b of the radical- chain mechanism for the autoxidation of a fatty acid hydrocarbon chain, you arrive at the following net equation: H I —CH2CH=CH—CH— + 0—0 • » Section of a fatty acid Oxygen hydrocarbon chain O—O—H I —CH2CH=CH—CH— A hydroperoxidearrow_forward2305032118517 CHO H-C-OH H-C-OH H₂C₂OH CHO Ho-C-tt A D-aldotetrose yields a mixture of the two compounds shown below when subjected to Kiliani - Fischer Chain extension synthesis. What is the Structure of the aldotetrose? 3 CH₂OH Compound I H-C-OH CHO HO-C-H Ho C-H сно HO-C-H WOO Н-С-он H-C-OH CH ₂ OH Compound II CHO H² C-OH HO-C-H CH₂OH CH ₂ OH CH₂OH CA) (B) (D) (E) Both choices (B) and (C) could be the correct structure of D-erythrose. CHO H₂C-OH HC OH CH ₂ OH ctrl MAND optionarrow_forwardDraw the major product(s) for reaction of methyl pentanoate with: (2) Me2CHCH2CH2OH, H30*arrow_forward
- 13-18.) Given the structures of the naturally occurring aldoses, list at least three examples of the following: a) diastereomers b) epimers Three carbons Four carbons Five carbons H--OH H--OH HO--H HO--H но-н H--OH ČH,OH H-C-OH HO--H H--OH H--OH ČH,OH -Erythrose D-Threose HO--H H-C-OH H-C-OH H--OH H--OH ČH,OH H--OH H-C-OH ČH,OH CH,OH CH,OH ČH,OH »Glyceraldehyde D-Ribose D-Arabinose D-Xylose D-Lyxose Six carbons H-C-OH HO-C-H H-C-OH HO--H HO-C-H H-C-OH HO-C-H H--OH HO-C-H H-C-OH HO-C-H HO-H HO--H HO--H HO--H H--OH CH,OH H-OH HO-H H-OH H--OH H--OH H-C-OH H-OH CH,OH H--OH CH,OH H-C-OH H OH CH,OH H-C-OH H--OH CH,OH HO-C-H H--OH CH,OH H-C-OH H--OH CH,OH D-Galactose -Talose CH,OH D-Allose D-Altrose D-Glucose D-Mannose D-Gulose D-Idose D-Aldoses (a)arrow_forwardIn dilute aqueous base, (R)-glyceraldehyde is converted into an equilibrium mixture of (R,S)-glyceraldehyde and dihydroxyacetone. Propose a mechanism for this isomerization. СНО СНО CH,OH NaOH СНОН СНОН C=0 CH,OH CH,OH ČH,OH (R)-Glyceraldehyde (R,S)-Glyceraldehyde Dihydroxyacetonearrow_forwardPropose synthetic routes to synthesize the target molecule OH Propose possible products formed in the following reaction OMe +arrow_forward
- CF 3- Celecoxib -SO2NH2 Do the following synthesis reactions. ŞO₂NH₂ NHNH2 A +arrow_forwardA Mannich reaction puts a R R-NCH2 - group on the α-carbon of a carbon acid. Propose a mechanism for the reaction.arrow_forwardWhich one below better describes the synthesis of steroids? Steroids synthesis depends on a cascade of carbocations and hydride and methyl shifts on a diterpene Steroids synthesis depends on the reaction of terpenes making cyclohexanes Steroids synthesis depends on epoxidation of C-2 of a triterpene that triggers carbocation rearrangements and elimination Steroids synthesis depends on conversion of a triterpene -squalene into 4 cyclohexanes fused ringsarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you

 Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning


Introduction to General, Organic and Biochemistry
Chemistry
ISBN:9781285869759
Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:Cengage Learning
Lipids - Fatty Acids, Triglycerides, Phospholipids, Terpenes, Waxes, Eicosanoids; Author: The Organic Chemistry Tutor;https://www.youtube.com/watch?v=7dmoH5dAvpY;License: Standard YouTube License, CC-BY