
Organic Chemistry (9th Edition)
9th Edition
ISBN: 9780321971371
Author: Leroy G. Wade, Jan W. Simek
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 21.12, Problem 21.35P
Show how you would synthesize each compound, starting with an ester containing no more than eight carbon atoms. Any other necessary reagents may be used.
- a. Ph3C-OH
- b. (PhCH2)2CHOH
- c. PhCONH CH2 CH3
- d. Ph2CHOH
- e. PhCH2OH
- f. PhCOOH
- g. PhCH2COOCH(CH3)2
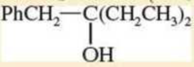
- h. HO-(CH2)8-OH
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Draw the product formed when phenylacetic acid (C6H5CH2COOH) is treated with each reagent. With some reagents, no reaction occurs.
a. NaHCO3
b. NaOH
c. SOCl2
d. NaCl
e. NH3(1equiv)
f. NH3, ∆
g. CH3OH, H2SO4
h. CH3OH, −OH
i. [1] NaOH; [2] CH3COCl
j. CH3NH2, DCC
k. [1] SOCl2; [2] CH3CH2CH2NH2 (excess)
l. [1] SOCl2; [2] (CH3)2; [2] (CH
Question 6
Give the reagents.
O 1. BH3, 2. H₂O2, NaOH
OmCPBA
O 1. Hg(OAc)2, H₂O 2. NaBH4
NaBH4
OH2, Pd/C
h
А.
Н Н
H-C-C-OH
Н Н
ос
О А
B
OD
D.
О Е
кн
Н
HIG
-Т
в.
Hс-с
Н
H-C-
H
Ошо
OH
Е.
H
An aldehyde is indicated by letter
H-C-N
Т
с.
Н-С-С-С-H
HOH
Н
Chapter 21 Solutions
Organic Chemistry (9th Edition)
Ch. 21.2F - Name the following carboxylic acid derivatives,...Ch. 21.4A - Prob. 21.2PCh. 21.4A - Prob. 21.3PCh. 21.4A - Prob. 21.4PCh. 21.5C - Prob. 21.7PCh. 21.6 - When ethyl 4-hydroxybutyrate is heated in the...Ch. 21.6 - Propose a mechanism for the following ring-opening...Ch. 21.6 - Prob. 21.15PCh. 21.7B - Prob. 21.16PCh. 21.7C - Prob. 21.19P
Ch. 21.7C - Prob. 21.20PCh. 21.7C - Prob. 21.21PCh. 21.7D - Prob. 21.22PCh. 21.7D - The mechanism for acidic hydrolysis of a nitrile...Ch. 21.8A - Prob. 21.24PCh. 21.8C - Prob. 21.25PCh. 21.9 - Prob. 21.26PCh. 21.9 - Prob. 21.27PCh. 21.9 - Prob. 21.28PCh. 21.10 - Draw a mechanism for the acylation of anisole by...Ch. 21.10 - Prob. 21.30PCh. 21.11 - Prob. 21.31PCh. 21.11 - Prob. 21.32PCh. 21.12 - Problem 21-33 Propose a mechanism for the...Ch. 21.12 - Suggest the most appropriate reagent for each...Ch. 21.12 - Show how you would synthesize each compound,...Ch. 21.13 - Prob. 21.36PCh. 21.13 - Prob. 21.37PCh. 21.14 - Prob. 21.38PCh. 21.14 - Prob. 21.39PCh. 21.16 - Prob. 21.40PCh. 21.16 - Prob. 21.41PCh. 21 - Prob. 21.42SPCh. 21 - Give appropriate names for the following...Ch. 21 - Predict the major products formed when benzoyl...Ch. 21 - Predict the products of the following reactions....Ch. 21 - Prob. 21.46SPCh. 21 - Prob. 21.47SPCh. 21 - Prob. 21.48SPCh. 21 - Propose mechanisms for the following reactions.Ch. 21 - Prob. 21.51SPCh. 21 - An ether extraction of nutmeg gives large...Ch. 21 - Prob. 21.53SPCh. 21 - Show how you would accomplish the following...Ch. 21 - Prob. 21.55SPCh. 21 - Prob. 21.56SPCh. 21 - Prob. 21.57SPCh. 21 - Prob. 21.58SPCh. 21 - Prob. 21.59SPCh. 21 - Explain this curious result. What does this...Ch. 21 - Prob. 21.61SPCh. 21 - Prob. 21.62SPCh. 21 - Prob. 21.63SPCh. 21 - A chemist was called to an abandoned aspirin...Ch. 21 - Prob. 21.67SPCh. 21 - The IR spectrum, 13ONTVTR spectrum, and 1HNMR...Ch. 21 - Prob. 21.69SPCh. 21 - Prob. 21.70SPCh. 21 - Prob. 21.71SP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Which ester can be easily condensed with sodium ethoxide to produce B-keto ester? Ph-C-OEt CH3-CH, C-OEt В. CH,-CH-C-OEt С. CH, 0 ҫ Н D. °f" A. 8 ČH, Öarrow_forward0 NaBH₁ Ethanol A. Product A B. Product B C. Product C D. Product D E. Product E major product A OH B OH BH₁ FFF HO D HO E 12:42 AMarrow_forwardDraw the products formed when p-methylaniline (p-CH3C6H4NH2) is treated with each reagent. a. HCl b. CH3COCl c. (CH3CO)2O d. excess CH3I e. (CH3)2C = O f. CH3COCl, AlCl3 g. CH3CO2H h. NaNO2, HCl i. Part (b), then CH3COCl, AlCl j. CH3CHO, NaBH3CNarrow_forward
- 2. Rank the following from highest to lowest water solubility: ОН v ОН ОН да - 0 H обира AL H - мен H Нarrow_forwardAcetal and Hemiacetal formation: c. Acetal/hemiacetal CH 3 ty CH H3C [ a. hemiacetal H3C d. Cyclic hemiacetal Gyertych OH Esterification/Amidation: e. Fisher esterification OH + CH₂OH + H3C CH3 H OH + CH3OH H [H*] H₂SO4 H™ a. hemiacetal b.acetal + H₂Oarrow_forward3. Complete the following reactions by adding the major product. 2 Ph OEt 1. NaOEt HOEt 2. H+ workup 1. NaOCH3 HOCH 3 2. H+ workup за Ph CI EtO OEt Ph OEt 1. NaOEt HOEt 2. H+ workup 1. NaOEt HOEt 2. H+ workuparrow_forward
- Choose the correct product for the given reaction. CF3 HBr ? CF3 CF3 Br A. Br Br CF3 Br CF3 D. Br С. Br A B B. ооо оarrow_forward9. Part 1: Draw the structure for compound A. Part 2: Draw the structure for compound B. Part 3: Identify the required reagent(s) for step 1. Select the single best answer. A) NaOH, H2O B) NaNH2, NH3 C) H2SO4 D) NH3 Part 4:Identify the required reagents for step 3. Select the single best answer. A) Na, NH3 B) H2, Pd C) H2, Lindlar catalyst D) NaNH2, NH3arrow_forwardOH 15. CH3-C-H ÓCH, The compound above is an example of a(n) a. acetal b. ketal O c. hemiacetal d. hemiketal e. none of the abovearrow_forward
- Which of the following reactions are correct? Explain the answer. + H₂O + H₂O H₂SO4 H OH H OHarrow_forwardDraw the product formed when phenylacetaldehyde (CsH;CH,CHO) is treated with each reagent. Phenylacetaldehyde is partly responsible for the fragrance of the flowers of the plumeria tree, which is native to the tropical and subtropical Americas. e. Ph;P=CHCH3 f. (CH),CHNH2, mild acid g. (CH,CH)NH, mild acid h. CH,CH,OH (еxсess), H' а. NaBHa, CH,Oн b. [1] LIAIH4; (2] H20 c. [1] CH;MgBr; [2] H2O i. NH, mild acid d. NaCN, HCI j. HOCH,CH;OH, H*arrow_forwardH. An ester is produced by the reaction of an alcohol with a carboxylic acid. For each ester below, underline the alcohol part and circle the carboxylic acid part. Then, name each ester. CHỊCH,CH,CH,CH,-O-C-CH,CH,CH, H CH₂ CH₂ CH 1 CH3arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY
Lipids - Fatty Acids, Triglycerides, Phospholipids, Terpenes, Waxes, Eicosanoids; Author: The Organic Chemistry Tutor;https://www.youtube.com/watch?v=7dmoH5dAvpY;License: Standard YouTube License, CC-BY