
Interpretation:
The shape of the labelled atoms in the below structure of enanthotoxin needs to be determined.
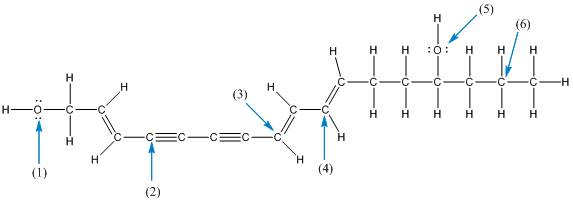
Concept introduction:
Shape of a molecule is determined by including only the bond pair not the lone pairs on the central atom while geometry includes both the bond pairs and lone pairs on the central atom. Valence shell electron pair repulsion theory or VSEPR theory used in chemistry as a model for the prediction of shape of various molecules by knowing the electron pairs on the central atom. There will be repulsion between the electron pairs present on central atom, so to minimize the repulsion they adopt an arrangement with minimum repulsion, thus determining molecule's shape. And by knowing the shape we can easily determine the bond angles.
The following table should be used while determining the shapes:
| Number of groups | Number of lone pairs | Shape | Bond angle | |
| 2 | 2 | 0 | Linear | |
| 3 | 3 | 0 | Trigonal planar | |
| 4 | 4 | 0 | Tetrahedral | |
| 4 | 3 | 1 | Trigonal pyramidal | |
| 4 | 2 | 2 | Bent |
Want to see the full answer?
Check out a sample textbook solution
Chapter 11 Solutions
General, Organic, and Biological Chemistry - 4th edition
- This question has multiple parts. Work all the parts to get the most points. For the reaction below: MgBr 1. ether 2. H30* Next Partarrow_forward#13.10arrow_forwardReset Help is polar 1. The main reason why CH3 OH has a higher vapor pressure at a given temperature when does not exhibit dispersion compared to CH3CH2CH2OH is that CH3 OH forces 2. The main reasons why CH4 has a higher vapor pressure at a given temperature when compared does not exhibit hydrogen to CH3CI is that CH4 and bonding has a smaller molar mass 3. The main reasons why H2 CO has a higher vapor pressure at a given temperature when does not exhibit dipole-dipole compared to CH3OH is that H2CO and forcesarrow_forward
- 10:33 Question 11 of 18 Select to Draw mCPBA a Submit me 1.0₂ 2. Zn/H₂O* Select to Drawarrow_forwardStudy guide answer is 4.77 for part 2 please show steps thanksarrow_forwardWhat type(s) of intermolecular forces are expected between CH3CH,CH2CH2CH2CH3 molecules? H H H H H H- H- H. H H H H. Indicate with a Y (ves) or an N (no) which apply. dipole forces induced dipole forces hydrogen bonding Submit Answer Try Another Version 2 item attempts remainingarrow_forward
- #10 help ,I got it wrongarrow_forwardDraw stable compounds using only the following elements. Do not make any coordinate covalent bonds A)Phosphorus and Fluorine B)Carbon and sulfur C)Aluminum and Oxygenarrow_forwardInstruction: Mr. Delphi is an elf known for being very creative by mixing different colors to his clothes. Color Mr. Delphi using the key below. Select the strongest force at work between the molecules. Color my Intermolecular Force lonic Bond- yellow Hydrogen bond-blue lon-dipole- black Dipole-dipole-green Dispersion force- red *hands, face, ears, nose = brown NO2 KBr in H₂O CaCl₂ CH3CH2CH3 HCN NH3 H3O CH4 NaCl He H₂O CO HI LiBr NaCl in H₂Oarrow_forward
- Practice Draw the structure of the following compounds. 1. bicyclo [4.2.1]nona-4,7-dien-2-ol 2. 5-bromo-7,7-dimethylbicyclo [2.2.1]heptan-2-one 3. 3-chloro-2,5-dimethylbicyclo[4.3.0]nonan-8-one 4. 6-ethoxy-1,2-dimethylspiro [2.4]heptane-5-carbaldehydearrow_forwardThis question has multiple parts. Work all the parts to get the most points. For the reaction below: NHarrow_forwardWhat is the specific feature of N, O, and F that causes them to play a role in hydrogen bonding? N, O, and F are all three atoms with electronegativity. When hydrogen is bonded to one of these atoms an especially dipole is created. Submit Answer Retry Entire Group No more group attempts remainarrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





