
Chemistry for Engineering Students
4th Edition
ISBN: 9781337398909
Author: Lawrence S. Brown, Tom Holme
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 7, Problem 7.93PAE
7.93 An unknown metal M forms a chloride with the formula 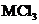 This chloride compound was examined and found to have a trigonal pyramidal shape. Draw a Lewis structure for
This chloride compound was examined and found to have a trigonal pyramidal shape. Draw a Lewis structure for 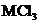 that is consistent with this molecular geometry. Use your structure to propose an identity for the metal M and explain how you have nude your choice.
that is consistent with this molecular geometry. Use your structure to propose an identity for the metal M and explain how you have nude your choice.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Boranes are a class of chemical compounds that undergo combustion reactions that are very similar to the reactions of hydrocarbons but they release more heat. If they were not so expensive, they would be considered for rocket fuel.
The structure of diborane is shown below. It “breaks rules” in terms of its Lewis structure. The “rule breaking” part is referred to as a “banana bond”.
a) Explain why it breaks rules.
b) Using the combustion of a hydrocarbon as your guide, predict the equation for the combustion of diborane. Balance the equation you predict.
6 7
8.
6.
10 11
12
13
13
CER: A chemist analyzed four different substances and obtained the given data below. Three of the four
substances are known. For the unknown substance, determine whether it utilizes ionic or covalent bonds. Use
evidence from the given data to support your claim. Explain your reasoning by referring to common properties
of ionic compounds.
目
Compound
Formula
Melting
Point
Physical
Appearance
Bolling Polnt
Solubility
Conductivity
Bond Type
White, big
crystals;
Not easily
Conducts
Calcium
Chloride
Soluble in
electricity
when
dissolved
CaCl,
782 °C
1936 °C
Ionic
water
crushed
White, small
crystals;
Easily
crushed
Does not
N/A,
Starts to
Soluble in
conduct
Covalent
Sucrose
C„H,,O,,
186 °C
water
burn
electricity
Does not
Slightly
soluble in
N/A,
White
conduct
Covalent
Acetaminophen
C,H,NO,
170 °C
Decomposes
powder
electricity
water
Conducts
White, small
crystals;
Easily
crushed
Soluble in
electricity
?
when
800 °C
1465 °C
water
Unknown
Unknown
dissolved
Decide whether these proposed Lewis structures are reasonable.
proposed Lewis structure
:Z:
: 0:
: 0:
:0
N=N
0=C=0
: 0:
]
- C.
—
: 0:
0:
Is the proposed Lewis structure reasonable?
Yes.
No, it has the wrong number of valence electrons.
The correct number is:
No, it has the right number of valence electrons but doesn't satisfy the
octet rule.
The symbols of the problem atoms are:*
Yes.
No, it has the wrong number of valence electrons.
The correct number is:
No, it has the right number of valence electrons but doesn't satisfy the
octet rule.
The symbols of the problem atoms are:* 0
Yes.
No, it has the wrong number of valence electrons.
The correct number is:
No, it has the right number of valence electrons but doesn't satisfy the
octet rule.
The symbols of the problem atoms are:
*
* If two or more atoms of the same element don't satisfy the octet rule, just enter the chemical symbol as many
times as necessary. For example, if two oxygen atoms don't satisfy the octet rule, enter "0,0".
Ś
Chapter 7 Solutions
Chemistry for Engineering Students
Ch. 7 - List some factors influencing the biocompatibility...Ch. 7 - • use electron configurations to explain why...Ch. 7 - • describe die energy changes in the formation of...Ch. 7 - • define electronegativity and state how...Ch. 7 - • identify or predict polar, nonpolar, and ionic...Ch. 7 - • write Lewis electron structures for molecules or...Ch. 7 - • describe chemical bonding using a model based on...Ch. 7 - • explain how hybridization reconciles observed...Ch. 7 - • predict the geometry of a molecule from its,...Ch. 7 - • use models (real or software) to help visualize...
Ch. 7 - • explain the formation of multiple bonds in terms...Ch. 7 - • identify sigma and pi bonds in a molecule and...Ch. 7 - Define the term biocompatibility.Ch. 7 - List some properties associated with biomaterials...Ch. 7 - Prob. 7.3PAECh. 7 - Prob. 7.4PAECh. 7 - Prob. 7.5PAECh. 7 - Prob. 7.6PAECh. 7 - Why is the ion not found in nature?Ch. 7 - Why do nonmetals tend to form anions rather than...Ch. 7 - Prob. 7.9PAECh. 7 - 7.10 Arrange the members of each of the following...Ch. 7 - 7.11 Arrange the following sets of anions in order...Ch. 7 - 7.12 Which pair will form a compound with the...Ch. 7 - 7.13 Figure 7-2 depicts the interactions of an ion...Ch. 7 - 7.14 Describe the difference between a covalent...Ch. 7 - 7.15 Covalently bonded compounds tend to have much...Ch. 7 - Prob. 7.16PAECh. 7 - 7.17 Coulombic forces are often used to explain...Ch. 7 - 7.18 In terms of the strengths of the covalent...Ch. 7 - 7.19 If the formation of chemical bonds always...Ch. 7 - 7.20 Draw the Lewis dot symbol for each of the...Ch. 7 - 7.21 Theoretical models for the structure of...Ch. 7 - 7.22 Use Lewis dot symbols to explain why chlorine...Ch. 7 - 7.23 Define the term lone pair.Ch. 7 - 7.24 How many electrons are shared between two...Ch. 7 - 7.25 How does the bond energy of a double bond...Ch. 7 - 7.26 How is electronegativity defined?Ch. 7 - 7.27 Distinguish between electron affinity and...Ch. 7 - 7.28 Certain elements in the periodic table shown...Ch. 7 - 7.29 When two atoms with different...Ch. 7 - 7.30 The bond in HF is said to be polar, with the...Ch. 7 - 7.31 Why is a bond between two atoms with...Ch. 7 - Prob. 7.32PAECh. 7 - 7.33 In each group of three bonds, which bond is...Ch. 7 - Prob. 7.34PAECh. 7 - 7.35 Which one of the following contains botb...Ch. 7 - Prob. 7.36PAECh. 7 - 7.37 Draw the Lewis structure for each of the...Ch. 7 - 7.38 Draw a Lewis structure for each of the...Ch. 7 - Prob. 7.39PAECh. 7 - 7.40 Why is it impossible for hydrogen to be the...Ch. 7 - Prob. 7.41PAECh. 7 - 7.42 Draw resonance structure for (a) (b) and (c)Ch. 7 - Prob. 7.43PAECh. 7 - Prob. 7.44PAECh. 7 - Prob. 7.45PAECh. 7 - 7.46 Consider the nitrogen-oxygen bond lengths in...Ch. 7 - 7.47 Which of the species listed has a Lewis...Ch. 7 - 7.48 Identify what is incorrect in the Lewis...Ch. 7 - 7.49 Identify what is incorrect in the Lewis...Ch. 7 - 7.50 Chemical species are said to be isoelectronic...Ch. 7 - 7.51 Explain the concept of wave interference in...Ch. 7 - 7.52 How does orbital overlap explain the buildup...Ch. 7 - 7.53 How do sigma and pi bonds differ? How are...Ch. 7 - 7.54 CO , CO2 , CH3OH , and CO32 , all contain...Ch. 7 - 7.55 Draw the Lewis dot structure of the following...Ch. 7 - 7.56 Draw the Lewis dot structures of the...Ch. 7 - 7.57 What observation about molecules compels us...Ch. 7 - Prob. 7.58PAECh. 7 - 7.59 What type of hybrid orbital is generated by...Ch. 7 - 7.60 What type of hybridization would be expected...Ch. 7 - 7.61 What hybrid orbitals would be expected for...Ch. 7 - 7.62 What type of hybridization would you expect...Ch. 7 - 7.63 What physical concept forms the premise of...Ch. 7 - 7.64 Predict the geometry of the following...Ch. 7 - Prob. 7.65PAECh. 7 - Prob. 7.66PAECh. 7 - Prob. 7.67PAECh. 7 - 7.68 Give approximate values for the indicated...Ch. 7 - 7.69 Propene has the chemical formula Describe the...Ch. 7 - Prob. 7.70PAECh. 7 - Prob. 7.71PAECh. 7 - 7.72 How does an MSN differ from amorphous silica...Ch. 7 - Prob. 7.73PAECh. 7 - 7.74 In a lattice, a positive ion is often...Ch. 7 - 7.75 Use the concept of lattice energy to...Ch. 7 - Prob. 7.76PAECh. 7 - Prob. 7.77PAECh. 7 - Prob. 7.78PAECh. 7 - Prob. 7.79PAECh. 7 - Prob. 7.80PAECh. 7 - Prob. 7.81PAECh. 7 - Prob. 7.82PAECh. 7 - Prob. 7.83PAECh. 7 - 7.84 Which of the following molecules is least...Ch. 7 - 7.85 Consider the molecule whose structure is...Ch. 7 - 7.86 Nitrogen triiodide, NI3(s) , is unstable and...Ch. 7 - 7.87 Nitrogen is capable of forming single,...Ch. 7 - 7.88 The N5+ cation has been synthesized and...Ch. 7 - Prob. 7.89PAECh. 7 - Prob. 7.90PAECh. 7 - 7.91 A Lewis structure for the oxalate ion is...Ch. 7 - Prob. 7.92PAECh. 7 - 7.93 An unknown metal M forms a chloride with the...Ch. 7 - Prob. 7.94PAECh. 7 - Prob. 7.95PAECh. 7 - 7.96 Consider the hydrocarbons whose structures...Ch. 7 - 7.97 Consider the structure shown below for as...Ch. 7 - Prob. 7.98PAECh. 7 - Prob. 7.99PAECh. 7 - Prob. 7.100PAECh. 7 - 7.101 Lead selenide nanocrystals may provide a...Ch. 7 - Prob. 7.102PAECh. 7 - Prob. 7.103PAECh. 7 - 7.104 Hydrogen azide, HN3 , is a liquid that...Ch. 7 - Prob. 7.105PAECh. 7 - Prob. 7.106PAECh. 7 - 7.107 How do the Lewis symbols for C, Si, and Ge...Ch. 7 - Prob. 7.108PAECh. 7 - Prob. 7.109PAE
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Draw Lewis diagrams for the following compounds. In the formula the symbol of the central atom is given first. (Hint: The valence octet may be expanded for the central atom.) (a) PF5 (b) SF4 (c) XeO2F2arrow_forwardMixing SbCl3 and GaCl3 in a 1:1 molar ratio (using liquid sulfur dioxide as a solvent) gives a solid ionic compound of empirical formula GaSbCl6 . A controversy arises over whether this compound is (SbCl2+)(GaCl4) or (GaCl2+)(SbCl4) . (a) Predict the molecular structures of the two anions. (b) It is learned that the cation in the compound has a bent structure. Based on this fact, which formulation is more likely to be correct?arrow_forward7.30 The bond in HF is said to be polar, with the hydrogen carrying a partial positive charge. For this to be true, the hydrogen atom must have less than one electron around it. Yet the Lewis dot structure of HF attributes two electrons to hydrogen. Draw a picture of the electron density distribution for HF and use it to describe how the hydrogen atom can carry a partial positive charge. How can these two models of the HF bond (the electron density and the Lewis structure) seem so different and yet describe the same thing?arrow_forward
- State the process of Predicting geometry from a valid Lewis structure ?arrow_forwardDirections: Look for at least 3 examples of covalent compounds that can be found in nature or used in everyday life. Include the following information: a. Brief information about the use of the covalent compound b. Chemical formula and chemical name of the covalent compound c. Lewis Structure of the compound d. Brief explanation if it follows the Octet Rulearrow_forward1. A) Predict formulas for all compounds that you might reasonably expect to form between each of the following pairs of elements based upon the Pairing Rule. Clearly show all reasoning. a) Se and F b) Sn and P c) In B) For each of the pairs of elements above, identify which compound, if any, satisfies the Octet Rule for all atoms. and Oarrow_forward
- Describe the difference between a full Lewis structure and bond-line notation. What changes? Why is it easier to write?Dontarrow_forwardNeed help I dont think I'm doing it correct describing the connectivity of the atoms by naming the central atom and those atoms bonded to it; pointing out and stating the value of the bond angles in degrees; naming the geometrical shape of the molecule surrounding the central atom; discussing the polarity of the molecule Compare and contrast the different types of bonds formed in molecules by writing Lewis structures and discussing the concepts of electronegativity, bond polarity, dipole moments and VSEPR theory.arrow_forward1. State Whether following statements are true (T) or False (F) in the space provided. a) Electron geometry and molecular geometry is not the same for all molecules. b) The enthalpy of neutralization of strong acid -strong base reaction is a constant. c) In the ideal gas law equation, the unit of temperature is in Kelvin (K). d) In an endothermic reaction heat is released from the system to the surroundings. e) Both AgCl and PbCl2 precipitates have the same color. contained in a calorimeter. When thermal equilibrium is reached, the temperature of the water is measured to be 30.6 °C. What is the specific heat of the metal? (10 pts) (heat capacity of water is 4.18 J/g°C) GHIZA 1 WLO TREICONAY aducation.ti.comarrow_forward
- A carbon oxygen bond is more polar than a carbon oxygen double bond. As a result, a Lewis structure for the carbonate ion has a slight molecular dipole. However, experimental evidence has clearly demonstrated that the carbonate ion has no net molecular dipole. Explain how a Lewis structure for carbonate can have a dipole and yet the actual molecule does not.arrow_forwardWriting Lewis Structures Many Lewis structures can be drawn by inspection but a system helps to draw the more complex ones. One approach is too : 1. Draw a skeleton of the major (non-hydrogen) atoms with single bonds ( lines) between each one. Several general considerations will help produce a valid skeletal diagram. normal covalency number or number of bonds that each atom forms. Consider the For the major atoms these are : Element Bond(s) HYDROGEN 1 BORON 3 ALUMINUM 3 CARBON | 4 NITROGEN 3 OXYGEN 2 HALOGENS 1 Also note that hydrogen, boron, aluminum and the halogens seldom form multiple bond.arrow_forwardIII. Determine the molecular geometry of the encircled atom in the following organic molecules by giving the required information in the table below: (Type your answers except for the Lewis structure. Draw the Lewis structure in a sheet of paper, scan copy or shot a picture of it and paste it next page.) LEWIS ELECTRON PAIR MOLECULAR MOLECULE STRUCTURE GEOMETRY SHAPE (4) CHs-CH-CH2-CNH-CH3 CH2CH3 (5) ATOM' 'ALENCE ELECTRON NO. OF BONDING PAIRS NO. OF NON- BONDING PAIRS TOTAL ELECTRON GENERAL FORMULA BOND ANGLE HYBRID ORBITALarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning
Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning
Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning
 World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co

Chemistry for Engineering Students
Chemistry
ISBN:9781337398909
Author:Lawrence S. Brown, Tom Holme
Publisher:Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:9781305079113
Author:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:Cengage Learning


World of Chemistry, 3rd edition
Chemistry
ISBN:9781133109655
Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:Brooks / Cole / Cengage Learning

Chemistry: Matter and Change
Chemistry
ISBN:9780078746376
Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:Glencoe/McGraw-Hill School Pub Co
Types of bonds; Author: Edspira;https://www.youtube.com/watch?v=Jj0V01Arebk;License: Standard YouTube License, CC-BY