
Concept explainers
(a)
Interpretation:
The mechanism for the given reaction should be determined.
Concept introduction:
Nucleophile: Nucleophiles are electron rich compounds which donates electrons to electrophilic compounds which results in bond formation.
Nucleophilic nature depends on the negative charge present in the molecule, the solvent in which it present and the electronegativity of the atom.
Electrophile: Electrophiles are electron deficient compounds which accepts electrons from nucleophiles that results in bond formation.
The curved arrows are generally used to indicate the flow of electrons present in the reaction.
Addition Reaction: It is defined as chemical reaction in which two given molecules combines and forms product. The types of addition reactions are electrophilic addition, nucleophilic addition, free radical additions and cycloadditions. Generally, compounds with carbon-hetero atom bonds favors addition reaction.
In addition reaction of
Oxidation Reaction: It involves loss of electrons, addition of oxygen atoms or removal of hydrogen atoms.
Oxidizing Reagents: The chemical agents used to add oxygen or remove hydrogen which finally reduced on oxidizing the other compound.
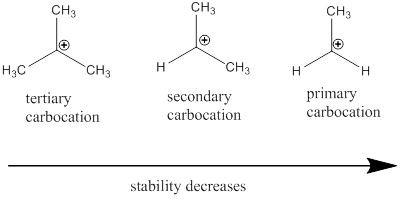
Carbocation: it is carbon ion that bears a positive charge on it.
Carbocation stability order:
(b)
Interpretation:
The rate determining step for the given reaction should be determined.
Concept introduction:
Carbocation: it is carbon ion that bears a positive charge on it.
Carbocation stability order:

Rate determining step: The slowest step in the reaction is called rate determining step. The actual
(c)
Interpretation:
The electrophile present in the first step of given reaction should be determined.
Concept introduction:
Electrophile: Electrophiles are electron deficient compounds which accepts electrons from nucleophiles that results in bond formation.
Chemical reaction involves bond making and breaking of two or more reactants in order to attain products from the reactants.
The curved arrows are generally used to indicate the flow of electrons present in the reaction.
(d)
Interpretation:
The nucleophile present in the first step of given reaction should be determined.
Concept introduction:
Nucleophile: Nucleophiles are electron rich compounds which donates electrons to electrophilic compounds which results in bond formation.
Nucleophilic nature depends on the negative charge present in the molecule, the solvent in which it present and the electronegativity of the atom.
(e)
Interpretation:
The electrophile present in the second step of given reaction should be determined.
Concept introduction:
Electrophile: Electrophiles are electron deficient compounds which accepts electrons from nucleophiles that results in bond formation.
Chemical reaction involves bond making and breaking of two or more reactants in order to attain products from the reactants.
The curved arrows are generally used to indicate the flow of electrons present in the reaction.
(f)
Interpretation:
The nucleophile present in the second step of given reaction should be determined.
Concept introduction:
Nucleophile: Nucleophiles are electron rich compounds which donates electrons to electrophilic compounds which results in bond formation.
Nucleophilic nature depends on the negative charge present in the molecule, the solvent in which it present and the electronegativity of the atom.
Trending nowThis is a popular solution!

Chapter 6 Solutions
Organic Chemistry
- Which factors would favor an SN2 reaction? Choose one or more: A. a strong nucleophile B. a good leaving group C. a high concentration of nucleophilearrow_forwardCan you match best nucleophile / conditions from the list that will give a successful hydrolysis reaction for each electrophile? (there's one best nucleophile / conditions for each electrophile) <List for Nucleophile / condition> a. LiAlH4; hydronium work-up b. PCC c. NaH d. CrO3 e. HOEt f. H3O+ or OH- g. H2O h. NaBH4;hydronium work-up Electrophile 1. Acid chloride 2. Acetic anhydride 3. Ester 4. Amide I'm asking again on bartleby because pther tutor gave me the right answer because I think I didn't make my question clear enough.arrow_forwardWhat mechanism is followed by Reaction 1 and 2? Possible Ans: Electrophilic addition, Electrophilic substitution, Radical substitution, Rearrangement What starting material is needed for reaction 1? a. Benzene b. Pent-2-ene c. Pent-2-yne d. Pentanearrow_forward
- For each alkyl halide and nucleophile: [1] Draw the product of nucleophilic substitution; [2] determine the likely mechanism (SN1 or SN2) for each reaction. a. CH;OH CH;CH20- Br b. "SH d. + CH;OH Brarrow_forwardWhich best describes this transformation? Meo,C CO,Me Et NaOMe (2 equivalents) MeOH Et H,N NH2 HN NH Et Et A. Two intermolecular nucleophilic acyl substitutions B. Two intramolecular nucleophilic acyl substitutions C. An intramolecular followed by an intermolecular nucleophilic acyl substitutions D. An intermolecular followed by an intramolecular nucleophilic acyl substitutionsarrow_forward3. a. Draw the major product that results from the nitration of anisole (shown below). OCH 3 anisole b. What reagents would be needed? c. What is the actual electrophile in this reaction (be specific)? Toisenons ontzi abouomo 20 d. How does the rate of this reaction compare to the nitration of benzene?arrow_forward
- Which member in each pair is a better leaving group? a. H2O or HO- b. NH3 or H2O c. H2O or H2S d. HO- or HS- e. I- or Br-. f. Cl- or Brarrow_forward5. Predict the major product of the following reactions. A. + HBr В. HBr C. + H Br 6. Use curved arrows to show to show the mechanism of the reaction C above. Nucleophiles and Electrophiles 7. Label the electrophile and nucleophile in each step of the mechanism for question 6.arrow_forwardThe reaction will proceed with mechanism because the secondary alkyl halide react with nucleophile. + NaOCH3 Br CH3 O a. SN2, strong O b. SN1, strong O c. SN1, weak O d. SN2, weakarrow_forward
- Draw the product of each reaction. a. + b. + C. d. + H2SO4 H2SO4 OH H2SO4 OH H2SO4arrow_forwardChoose the correct answer and briefly explain yo a. The most reactive alkyl halide in a E1 reaction b. The most reactive molecule in a S№2 reaction: i A B c. The most reactive alkyl bromide in a S№1 react Br Br A Barrow_forward8. Which of the following compounds is capable of undergoing an SN1 or SN2 substitution reaction? (more than one answer may be correct, circle all correct answers) OH2arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





