
Concept explainers
Interpretation:
The given questions under given set of conditions should be answered.
Concept introduction:
Nucleophile: Nucleophiles are electron rich compounds which donates electrons to electrophilic compounds which results in bond formation.
Nucleophilic nature depends on the negative charge present in the molecule, the solvent in which it present and the electronegativity of the atom.
Electrophile: Electrophiles are electron deficient compounds which accepts electrons from nucleophiles that results in bond formation.
The curved arrows are generally used to indicate the flow of electrons present in the reaction.
Addition Reaction: It is defined as chemical reaction in which two given molecules combines and forms product. The types of addition reactions are electrophilic addition, nucleophilic addition, free radical additions and cycloadditions. Generally, compounds with carbon-hetero atom bonds favors addition reaction.
In addition reaction of
Oxidation Reaction: It involves loss of electrons, addition of oxygen atoms or removal of hydrogen atoms.
Oxidizing Reagents: The chemical agents used to add oxygen or remove hydrogen which finally reduced on oxidizing the other compound.
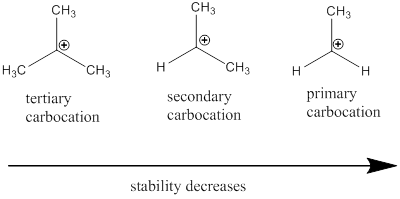
Carbocation: it is carbon ion that bears a positive charge on it.
Carbocation stability order:
Want to see the full answer?
Check out a sample textbook solution
Chapter 6 Solutions
Organic Chemistry
- Draw the product that would form when 4-methyl-2-pentene reacts with bromine. • You do not have to consider stereochemistry. • You do not have to explicitly draw H atoms. • In cases where there is more than one answer, just draw one. / Sn [F ? ChemDoodlearrow_forwardDraw the structure(s) of the major organic product(s) of the following reaction. s ● PPh₂ • You do not have to consider stereochemistry. • You do not have to explicitly draw H atoms. Do not include lone pairs in your answer. They will not be considered in the grading. • Omit the phosphine oxide from your answer. . If no reaction occurs, draw the organic starting material. · • Draw one structure per sketcher. Add additional sketchers using the drop-down menu in the bottom right corner. Separate multiple products using the + sign from the drop-down menu. + 99-81 ***11 / / | ? (10 ChemDoodle Sn [Farrow_forwardAs we will learn in Section 13.12, many antioxidants–compounds that prevent unwanted radical oxidation reactions from occurring–are phenols, compounds that contain an OH group bonded directly to a benzene ring. a. Explain why homolysis of the O–H bond in phenol requires considerably less energy than homolysis of the O–H bond in ethanol (362 kJ/mol vs. 438 kJ/mol). b.Why is the C–O bond in phenol shorter than the C–O bond in ethanol?arrow_forward
- In halogenation reactions, why are halogens added on opposite sides of the compound? because the reaction proceeds via free radical and the pi cloud holding the electron are directed away from each other because of the formation of a halonium ion which sterically blocks one side of the compound thus the other side is open for attack because the halogen should occupy both sides to accommodate the pi clouds None of the above Two equivalents of HCl are added to an alkyne, where do you expect to see the Cl atom? The two Cl atoms will be distributed between the two Carbon atoms involved in the triple bond The two Cl atoms forms a bond with the less substituted carbon Both Cl atoms attach to one carbon, which is the more substituted one A mixture of products are formedarrow_forwardA benzene ring alters the reactivity of a neighboring group in the so-called “benzylic” position, similarly to how a double bond alters the reactivity of groups in the “allylic” position. Benzylic cations, anions, and radicals are all more stable than simple alkyl intermediates. a) Use resonance structures to show the delocalization of the positive charge, negative charge, and unpaired electron of the benzyl cation, anion, and radical.arrow_forwardWhy does acrylonitrile (A) react faster with 1,3-butadiene in a pericyclic reaction than methyl vinylether (B)? Check all that apply. CEN A O 1. The bond lenght between carbon and nitrogen is greater than the bond lenght between carbon and oxygen which brings the bulky cyano group further away from the carbon-carbon double bond. That reduces sterical O 2. The LUMO of acrylonitrile (A) is has a lower energy than the LUMO of methyl vinylether (B) which makes the reaction go faster O 3. The high electronegativity of the oxygen atom in ether B polarizes the carbon-carbon double bond which reduces the reaction rate. O 4. The boiling point of nitrile A is higher than the boiling point of ether B, therefore the reaction can be carried out at a higher temperature which accelerates the reaction. hindrance and accelerates the reaction.arrow_forward
- Electrophilic aromatic substitutions proceed in two stages. 1. Attack of the aromatic ring on the electrophile. 2. Regeneration of the stable aromatic system by loss of a positively charged unit, usually a proton. Br Br₂/FeBr3 Draw one resonance structure for the intermediate formed when the electrophile is attacked by benzene in the reaction above. • You do not have to consider stereochemistry. • You do not have to explicitly draw H atoms. • Do not include lone pairs in your answer. They will not be considered in the grading.arrow_forwardElectrophilic aromatic substitutions proceed in two stages. 1. Attack of the aromatic ring on the electrophile. 2. Regeneration of the stable aromatic system by loss of a positively charged unit, usually a proton. Br Br₂/FeBr3 Draw one resonance structure for the intermediate formed when the electrophile is attacked by benzene in the reaction above. • You do not have to consider stereochemistry. • You do not have to explicitly draw H atoms. • Do not include lone pairs in your answer. They will not be considered in the grading. (+) ChemDoodleⓇarrow_forwardA Question Completion Status: 6 10 13 14 15 21 25 A Moving to another question will save this response. Question 21 In the standard electrophilic addition of a hydrohalide (H-X) to an alkene, what can be said about the stereochemistry of the product? O The stereochemistry will be anti, since the sigma bond will rotate once the pi bond is broken O The stereochemistry will be syn, since the nucleophile will be generated on the same side of the alkene as the electrophile. O The stereochemistry will be random, since the carbocation generated is planar and attack can come from either side. O None of these, because it is not possible to generate a chiral center with stereochemistry through an electrophilic addition reaction A Moving to another question will save this response. 080 acerarrow_forward
- The two reactants shown below are combined to bring about a nucleophilic substitution reaction. [References] a + Nal C. Br e. I i. HI HỌ. g. HBr j. Na* k. Br h. H20 1. none Which letter designates the electrophilic carbon at which substitution occurs? (If no reaction occurs enter the letter corresponding to "none.") Which letter corresponds to the leaving group? Which letter designates the nucleophile?arrow_forwardShow how you might synthesize this compound from an alkyl bromide and a nucleophile in an SN2 reaction. / من • Use the wedge/hash bond tools to indicate stereochemistry where it exists. • Only draw the reactants. Separate multiple reactants using the + sign from the drop-down menu. • If there is more than one possible combination of alkyl bromide and nucleophile, draw only one combination. • Do not include counter-ions, e.g., Na+, I, in your answer. Submit Answer N3 ? ChemDoodle Retry Entire Group ▼n n [ ]# 8 more group attempts remainingarrow_forwarda.) What was the product of the first step (reaction involving HCl)? b.)What was the product of the second step (reaction involving NaOH)? If something else formed as the product of the first reaction, use that alternative structure as the reactant for this second reaction. c.)How would you change the reaction conditions (either step 1 or step 2) to ensure you obtained the planned final product as the major product.arrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
