
(a)
Interpretation:
The Lewis structure for
Concept Introduction:
Lewis structure is also known as Lewis dot diagrams or electron dot structures. The bond between atoms and lone pairs of electrons that is present in the molecule. Lewis structure represents each atom and their position in structure using the chemical symbol. Excess electrons forms the lone pair are given by pair of dots, and are located next to the atom.
(a)
Explanation of Solution
Oxygen is in Group 6A and Chlorine is in Group 7A and the valence electrons present in the
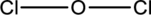
The two chlorine atoms connect with one Oxygen atom through single bonds.
Chlorine atoms attain octet by adding six electrons as dots in pairs.
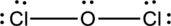
Complete the octet of the two chlorine atom uses
Put the last six electrons on Oxygen atom.
The correct Lewis structure of the
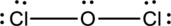
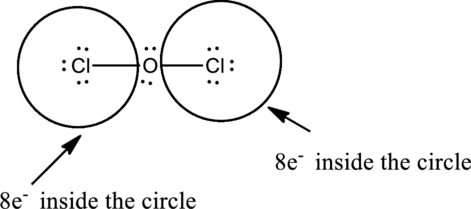
Oxygen has eight electrons four in the bonds and four as dots, hence the structure is complete.
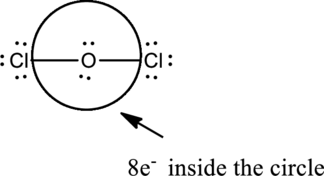
Hence, the total number of electrons can be counted as
(b)
Interpretation:
The Lewis structure for
Concept Introduction:
Refer part (a).
(b)
Explanation of Solution
Hydrogen atom is from Group one A and Oxygen atom is from group 6A, hence the valence electrons are
Complete the octet of the two chlorine atom uses
The incomplete Lewis structure of
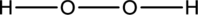
The correct Lewis structure of the

Hence, the total number of electrons can be counted as
(c)
Interpretation:
The Lewis structure for
Concept Introduction:
Refer part (a).
(c)
Explanation of Solution
The four Hydrogen atoms connect to boron with single bonds uses eight electrons. Boron atom is the central atom with the hydrogen atoms around it. So, the valence electron present in the
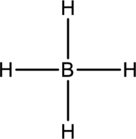
Boron atom must be the central atom with the four Hydrogen atoms bonded to it. Boron has eight electrons so, the structure is complete. Boron has eight electrons, and each Hydrogen atom has just two electrons, the structure clockwise, the total number of electrons can be counted
The correct Lewis structure of the
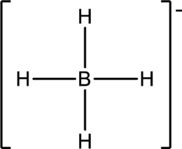
(d)
Interpretation:
The Lewis structure for
Concept Introduction:
Refer part (a).
(d)
Explanation of Solution
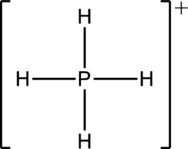
The four Hydrogen atoms connect to Phosphorous with single bonds uses eight electrons. Phosphorous atom is the central atom with the hydrogen atoms around it. So, the valence electrons present in the
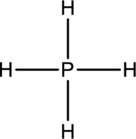
Phosphorous atom must be the central atom with the four Hydrogen atoms bonded to it. Phosphorous has eight electrons so, the structure is complete. It has eight electrons, and each Hydrogen atom has just two electrons, the structure clockwise, the total number of electrons can be counted
The structure is a
The correct Lewis structure of the
(e)
Interpretation:
The Lewis structure for
Concept Introduction:
Refer part (a).
(e)
Explanation of Solution
The five chlorine atoms connect to Phosphorous with single bonds uses ten electrons. Phosphorous atom is the central atom with the Chlorine atoms around it.
The number of valence electrons present in
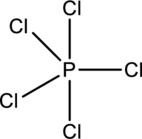
Chlorine atoms prefer making only one bond, and Phosphorous prefers to make three and five bonds. So use Phosphorous atom as central atom with the five Chlorine atoms around it.
Each Chlorine atom has three lone pair and one bond pair so it attains octet.
The correct Lewis structure of the
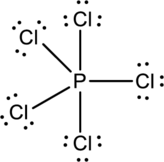
The total number of electrons can be counted as
Want to see more full solutions like this?
Chapter 6 Solutions
Chemistry: The Molecular Science
- Boric acid, B(OH)3, is an acid that acts differently from the usual Brønsted-Lowry acids. It reacts with water according to B(OH);(aq) + 2 H,0(€)2B(OH), (aq) + H;O* (aq) K, = 5.8 × 10¬10 (a) Draw Lewis structures for B(OH); and B(OH)4. Can these be described as Lewis acids or Lewis bases? (b) Calculate the pH of a 0.20 M solution of B(OH)3(aq).arrow_forwardWrite the Lewis structure for each of the following species,and indicate the structure of each: (a) SeO32 - ; (b) S2Cl2;(c) chlorosulfonic acid, HSO3Cl (chlorine is bonded tosulfur).arrow_forward4. (a) Draw the shape of the atomic valence orbitals formed by the overlaping of two fluoride 2p atomic orbitals. (b) Draw the molecular orbital diagrams for F2 and F2*. Identify their bond order and magnetic properties. (c) An unstable nucleus exhibit radioactivity. (i) Explain how the number of protons and neutrons in a radioactive nucleus can be used to predict its probable mode decay. (ii) Illustrate your answer in (i) with a schematic graph.arrow_forward
- Which of the following bonds are polar: (a) P—O; (b) S—F; (c) Br—Br; (d) O—Cl? Which is the more electronegative atom in each polar bond?arrow_forwardDraw the Lewis structure for (a) NO+ ion, (b) C2H4.arrow_forward(a) What are trihalomethanes (THMs)? (b) Draw the Lewisstructures of two example THMs.arrow_forward
- (a) Triazine, C3 H3 N3, is like benzene except that in triazineevery other C¬H group is replaced by a nitrogen atom.Draw the Lewis structure(s) for the triazine molecule. (b) Estimatethe carbon–nitrogen bond distances in the ring.arrow_forwardCompounds such as NaBH4, Al(BH4)3, and LiAlH4 are complex hydrides used as reducing agents in many syntheses. (a) Give the oxidation state of each element in these compounds. (b) Write a Lewis structure for the polyatomic anion in NaBH4, and predict its shape.arrow_forward(a) Triazine, C3 H3 N3, is like benzene except that in triazineevery other C¬H group is replaced by a nitrogen atom. Draw the Lewis structure(s) for the triazine molecule. (b) Estimatethe carbon–nitrogen bond distances in the ring.arrow_forward
- Compounds such as NaBH4, Al(BH4)3, and LiAlH4 are complexhydrides used as reducing agents in many syntheses.(a) Give the oxidation state of each element in these compounds.(b) Write a Lewis structure for the polyatomic anion in NaBH4, and predict its shape.arrow_forwardFor each of the following covalent bonds: (a) use the symbols δ+ and δ- to indicate the direction of polarity (if any).(a) C-F; (b) N-Br; (c) B-C; (d) Si-H(b) Rank the following covalent bonds in order of increasing polarity. (i) C-H, O-H, N-H; (ii) C-N, C-O, B-O; (iii) C-P, C-S, C-Narrow_forwardAcetylene 1C2H22 and nitrogen 1N22 both contain a triplebond, but they differ greatly in their chemical properties.(a) Write the Lewis structures for the two substances. (b) Byreferring to Appendix C, look up the enthalpies of formationof acetylene and nitrogen. Which compound is more stable?(c) Write balanced chemical equations for the completeoxidation of N2 to form N2O51g2 and of acetylene to formCO21g2 and H2O1g2. (d) Calculate the enthalpy of oxidationper mole for N2 and for C2H2 (the enthalpy of formationof N2O51g2 is 11.30 kJ>mol). (e) Both N2 and C2H2 possesstriple bonds with quite high bond enthalpies (Table 8.3).Calculate the enthalpy of hydrogenation per mole for bothcompounds: acetylene plus H2 to make methane, CH4;nitrogen plus H2 to make ammonia, NH3.arrow_forward
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
