
(a)
Interpretation:
For given named compound the formula has to be written.
Concept introduction:
Naming molecular compounds:
To named binary ionic compounds, first predict the positive ion and then negative ion. The positive ion as named as same element while negative ion taking the first part from its original name and ending with –ide.
Numerical prefix for naming compounds are,
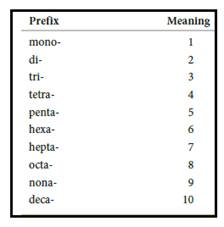
Figure 1
(b)
Interpretation:
For given named compound the formula has to be written.
Concept introduction:
Naming molecular compounds:
To named binary ionic compounds, first predict the positive ion and then negative ion. The positive ion as named as same element while negative ion taking the first part from its original name and ending with –ide.
Numerical prefix for naming compounds are,

Figure 1
(c)
Interpretation:
For given named compound the formula has to be written.
Concept introduction:
Naming molecular compounds:
To named binary ionic compounds, first predict the positive ion and then negative ion. The positive ion as named as same element while negative ion taking the first part from its original name and ending with –ide.
Numerical prefix for naming compounds are,
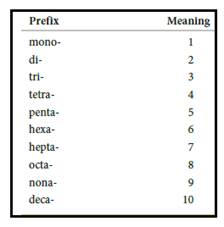
Figure 1
(d)
Interpretation:
For given named compound the formula has to be written.
Concept introduction:
Naming molecular compounds:
To named binary ionic compounds, first predict the positive ion and then negative ion. The positive ion as named as same element while negative ion taking the first part from its original name and ending with –ide.
Numerical prefix for naming compounds are,
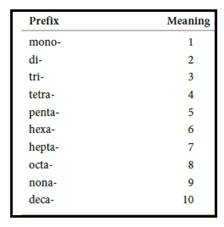
Figure 1
(e)
Interpretation:
For given named compound the formula has to be written.
Concept introduction:
Naming molecular compounds:
To named binary ionic compounds, first predict the positive ion and then negative ion. The positive ion as named as same element while negative ion taking the first part from its original name and ending with –ide.
Numerical prefix for naming compounds are,

Figure 1
Want to see the full answer?
Check out a sample textbook solution
Chapter 4 Solutions
General Chemistry: Atoms First
- Which of the following compounds has both ionic and covalent bond? (a) H2O (b) NH4Cl (c) NaCl (d) HClarrow_forwardWrite the formula of the compound that will be formed between these elements: (a) H and F (b) Cs and P (c) Al and Clarrow_forwardDetermine whether the following atoms will form an ionic compound or a molecular compound, and give the formula of the compound. (a) sodium and chloride (b) carbon and 4 hydrogen (c) magnesium and brominearrow_forward
- Write a formula for each ionic compound.(a) copper(II) bromide(b) silver nitrate(c) potassium hydroxide(d) sodium sulfate(e) potassium hydrogen sulfate(f) sodium hydrogen carbonatearrow_forwardWhat are likely formulas for the following molecules? Supply the missing digit. Choose 1 if the element appears in the formula as a single atom. Hydrogens are bonded to the preceeding atom in the formula. (a) CH,NH, (b) SH₂ (c) HOCI, 12345arrow_forwardWrite a formula for each of the following compounds:(a) silicon dioxide(b) silicon tetraiodide(c) silane(d) silicon carbide(e) magnesium silicidearrow_forward
- - Classify each compound as ionic or molecular. If it is ionic, determine whether the metal forms only one type of ion or more than one type of ion. (a) CoCl2 (b) CF4 (c) BaSO4 (d) NOarrow_forwardWhich of the following is an ionic compound? (A) (NH4)3PO4 (B) H3PO4 (C) SiCl4 (D) PCl5 (E) All of them arearrow_forwardPredict which of these compounds are ionic and which are covalent.(A) Ca3N2(B) Li2CO3(C) PCl5(D) NaOH(E) CH4(F) MgOarrow_forward
- Predict whether the following compounds are ionic or molecular:(a) KI, the compound used as a source of iodine in table salt(b) H2O2, the bleach and disinfectant hydrogen peroxide(c) CHCl3, the anesthetic chloroform(d) Li2CO3, a source of lithium in antidepressantsarrow_forwardEach of the following compounds contains a metal that can exhibit more than one ionic charge. Name these compounds:(a) Cr2O3(b) FeCl2(c) CrO3(d) TiCl4(e) CoO(f) MoS2arrow_forwardWrite formulas for the following anions: (a) fluoride (h) oxide (b) acetate (i) dichromate (c) iodide (j) hydrogen carbonate (d) carbonate (k) phosphate (e) sulfide (l) sulfate (f) nitrate (m) nitride (g) phosphide (n) chloridearrow_forward
 Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
