
Concept explainers
Interpretation:
The fraction of the receptor bonded with the hormone needs to be calculated.
Concept Introduction :
While considering the simple equilibrium binding of receptor to hormone the below equilibrium could be used:
Within this equilibrium, [HR] is representative of the hormone’s concentration receptor complex. [H] signifies the free hormone’s concentration while [R] signifies the noncomplexed receptor’s concentration. The equilibrium dissociation constant for this specific reaction could be found to be as:
Answer to Problem 9P
Explanation of Solution
The equilibrium equation can also be written as follows:
After substituting,
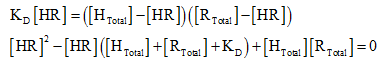
The quadratic equation to solve for the variables:
Substituting in the initial hormone and receptor concentration gives:
Since [HR] must be 0 when
To determine the fraction receptor bound divide [HR] by
These calculations can be repeated for when the concentration of receptor is decreased to 0.33 nM.
Taking the negative root again:
Finding the fraction receptor bound gives:
The fraction of the receptor bonded with the hormone is 0.496.
Want to see more full solutions like this?
- Insulin is a hormone that regulates blood sugar by binding to its receptor, insulin receptor tyrosine kinase. How does insulin's behavior differ from steroid hormone signaling, and what can you infer about its structure?arrow_forwardIf intracellular [ATP] = 5 mM, [ADP] = 0.5 mM, and [Pi] = 1.0 mM, calculate the concentration of AMP at pH 7 and 25°C under the condition that the adenylate kinase reaction is at equilibrium.arrow_forwardTrue or False. The binding affinity between a peptide agonist and its specific G protein coupled receptor involves certain types of chemical bonds. Explain your answer in detail.. ( please do not copy off of another source.. e.g chegg)arrow_forward
- True or False. The binding affinity between a peptide agonist and its specific G protein coupled receptor alone determines the sensitivity of a cell to the agonist peptide.arrow_forward1E In terms of binding adenylate kinase, the Kd for ATP is ~50 M and the KI for GMP-PCP is ~50 nM. Explain how this may be possible in terms of molecular interactions. Name 4 different types of molecular interactions that may contribute to the increased binding affinity exhibited by GMP-PCP. Please help me in detailsarrow_forwardEndocrine - Can I get the answer? I do not need explanation. 1. The â C peptideâ of the insulin prohormone can be used clinically to monitor endogenous insulin production. True False 2. HbA1c is a glycated hemoglobin in which a glucose molecule is covalently bound to the N-terminal valine of a hemoglobin subunit. True False 3. HbA1c is used to monitor blood glucose levels because hemoglobin is the only protein in blood that is covalently modified by glucose. True Falsearrow_forward
- PRESSURE OF A WHAT IS THE asmoTIC SOLUTION OF BOVINE insuLIN (MOLAR MASS, 5700g mol - 1 ) aT 18 °C IF 100.0 ML OF THE SOLUTION conTains 0.103 a OF THE INSULIN?arrow_forward2+ The activity of the Ca 2+ -ATPase is regulated reversibly under normal conditions to maintain homeostatic concentrations of Ca²- inside the sarcomere. However, in a rare genetic disorder, irreversible activation of the Ca 2+ -ATPase can occur. Assuming 37 °C, pH = 7.4, and the steadystate concentrations for ATP, ADP Pi, and Ca2+ (out) given in part (a), calculate the minimum [Ca2+] inside a sarcomere that has irreversibly activated Ca 2+ -ATPase (i.e., the Ca2+-ATPase activity is always “on”). Express your answer to two significant figures and include the appropriate units.arrow_forwardLigand binding to proteins may occur with varying strengths; some ligands bind tightly to proteins while others bind less tightly. The strengths of reversible binding are determined experimentally by varying concentrations of ligands, and measuring the saturation of the protein in the various ligand concentrations. One such laboratory study investigated the binding of a hormone to three different receptor proteins in the cell membrane. The data collected are shown in the table below: 1) Provide a brief explanation as to why ligand binding to proteins must be a reversible process. 2) Calculate the dissociation constant (Kd) for the hormone binding to each of the three proteins.arrow_forward
- 01 M Inbox (449) - krys. Start Later, Sleep. 8 Amazing Benefit... /SC The drug Ouabain inhibits the function of the sodium-potassium pump. Predict the short-term and long-term effects of Ouabain on the excitability (ability to be stimulated) of a neuron. Think about the effect this would have on the resting membrane potential. A- В I 三 三 X2 x² =三三arrow_forwardLigand binding to proteins may occur with varying strengths; some ligands bind tightly to proteins while others bind less tightly. The strengths of reversible binding are determined experimentally by varying concentrations of ligands, and measuring the saturation of the protein in the various ligand concentrations. One such laboratory study investigated the binding of a hormone to three different receptor proteins in the cell membrane. The data collected are shown in the table below 1) Which of the proteins demonstrate the tightest/strongest binding of the hormone? Which demonstrates the least tight binding of the hormone? What is the basis for your answer?arrow_forwardLigand binding and response. The following question involves the ligand binding to a receptor and the receptor's response to that ligand. What ligand concentration would be required for a full agonist with a KD of 8 nM to achieve a response of 0.75?arrow_forward
 BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax
Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax Biology: The Dynamic Science (MindTap Course List)BiologyISBN:9781305389892Author:Peter J. Russell, Paul E. Hertz, Beverly McMillanPublisher:Cengage Learning
Biology: The Dynamic Science (MindTap Course List)BiologyISBN:9781305389892Author:Peter J. Russell, Paul E. Hertz, Beverly McMillanPublisher:Cengage Learning


