
Concept explainers
Interpretation:
The
Concept Introduction:
A wave is a disturbance or variation that travels through a medium transporting energy without transporting matter. The wavelength is the distance between identical points on successive waves. The frequency is the number of waves that pass through any particular point in 1 second.
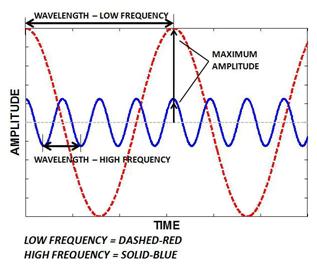
Figure1
The speed, wavelength and frequency of a wave are related by the equation:
To find: The frequency of light with wavelength of
Correct answer:
The frequency of light with wavelength of
Want to see the full answer?
Check out a sample textbook solution
Chapter 3 Solutions
Chemistry: Atoms First
- A particular transition of the rubidium atom emits light whose frequency is 3.84 1014 Hz. (Hz is the abbreviation for hertz, which is equivalent to the unit/s, or s1.) Is this light in the visible spectrum? If so, what is the color of the light? (See Figure 7.5.)arrow_forwardWhat is the wavelength of the electromagnetic radiation emitted from a hydrogen atom when the electron undergoes the transition n = 4 to n = 1? In what region of the spectrum does this line occur? (See Figure 7.5.)arrow_forward(a) Calculate the frequency and the wavelength of the line for the n = 6 to n = 4 transition. (b) Is this wavelength longer or shorter than that of the n = 7 to n = 4 transition?arrow_forward
- What is the wavelength of a proton traveling at a speed of 6.21 km/s? What would be the region of the spectrum for electromagnetic radiation of this wavelength?arrow_forwardLight of wavelength 345 nm shines on a piece of calcium metal. What is the speed of the ejected electron? (Light energy greater than that of the work function of calcium ends up as kinetic energy of the ejected electron. See Problem 7.75 for the definition of work function and its value for calcium.)arrow_forwardIn X-ray fluorescence spectroscopy, a material can be analyzed for its constituent elements by radiating the material with short-wavelength X rays, which induce the atoms to emit longer-wavelength X rays characteristic of those atoms. Tungsten, for example, emits characteristic X rays of wavelength 0.1476 nm. If an electron has an equivalent wavelength, what is its kinetic energy?arrow_forward
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning





