
Organic Chemistry: A Guided Inquiry
2nd Edition
ISBN: 9780618974122
Author: Andrei Straumanis
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 3, Problem 2E
Explain why the two molecules below cannot inter-convert without breaking the
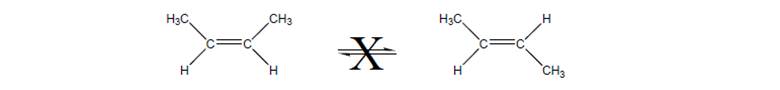
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Consider the T-system of the radical (a molecule with one
unpaired electron) shown below.
Note: the dot in this drawing represents an unpaired electron
What is the best description of the following n molecular
orbital (hint: consider the resonance structures of the radical)?
ㅇ
Note: each dot in this drawing represents a carbon nucleus
bonding and filled with two electrons
bonding and empty
non-bonding and contains one electron
antibonding and empty
antibonding and filled with two electrons
Assign hybridization state of each carbon, nitrogen, oxygen, halogen, and boron atom indicated with arrows in the molecules below.
Which of the following statements about the formation of hybrid orbitals is false?
O The shapes of hybrid orbitals are different from the shapes of atomic orbitals.
O Hybrid orbitals are a means to explain the formation of bonds consistent with the molecular shapes predicted by the VSEPR model.
O Hybrid orbitals are created by mixing an orbital on one atom with an orbital on a different atom.
O The formation of hybrid orbitals may require the promotion of an electron from an s orbital to ap orbital.
Chapter 3 Solutions
Organic Chemistry: A Guided Inquiry
Ch. 3 - Prob. 1CTQCh. 3 - What neutral atom is represented by the electron...Ch. 3 - Prob. 3CTQCh. 3 - Consider any one of the four identical hybrid...Ch. 3 - Prob. 5CTQCh. 3 - Prob. 6CTQCh. 3 - Prob. 7CTQCh. 3 - Prob. 8CTQCh. 3 - Prob. 9CTQCh. 3 - Prob. 10CTQ
Ch. 3 - On the left side of Figure 3.6, label the areas...Ch. 3 - Prob. 12CTQCh. 3 - Prob. 13CTQCh. 3 - Prob. 14CTQCh. 3 - Prob. 15CTQCh. 3 - Now consider the fully formed molecule on the...Ch. 3 - Prob. 1ECh. 3 - Explain why the two molecules below cannot...Ch. 3 - Prob. 3ECh. 3 - Consider the incomplete orbital representation of...Ch. 3 - Consider the following orbital representation of...Ch. 3 - Summarize how one determines the hybridization...Ch. 3 - Explain what is wrong with each of the following...Ch. 3 - Prob. 8ECh. 3 - Prob. 9ECh. 3 - Complete the following tables, and memorize their...Ch. 3 - Draw orbital representations of bonding in water...Ch. 3 - Draw electron configuration diagrams for carbon in...Ch. 3 - Prob. 13E
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- A student who missed this class needs to know how to predict the bond angles and shape of amolecule from looking at its bond-line representation. Write a concise but complete explanationfor this student.arrow_forwardDraw the delocalized molecular orbitals for the following molecule. Are both bonds of the triple bond involved in the delocalized orbitals?arrow_forwardAspartame is a compound that is 200 times sweeter than sugar and is used extensively (under the trade name NutraSweet) in diet soft drinks. The skeleton structure of the atoms in aspartame is (a) Complete the Lewis structure and give the number of and bonds in aspartame. (b) What is the hybridization about each carbon atom that forms a double bond with an oxygen atom? (c) What is the hybridization about each nitrogen atom?arrow_forward
- In how many sigma bonds does the highlighted atom participate?arrow_forwardDraw the valence bond diagram representing chemical bonding in the molecule of HCONH2 , showing atomic and hybridized orbitals as boxes, valence electrons as arrows in the boxes, and chemical bonds as lines connecting the boxes. Note that the lone pair on N occupies an unhybridized 2p orbital due to the contribution of resonance structure.arrow_forwardShow which of the following molecule will have dipole moment in each case and show their directions.arrow_forward
- Describe the orbitals that are mixing to make the bond that the thicker arrow is pointing to. (formatting is shown for the C:O double bond pointed to by the thinner arrow)arrow_forwardIn what type of orbital does each lone pair reside?arrow_forwardConsidering the molecule below, in what type of orbital does each lone pair of electrons on the oxygen atom reside?arrow_forward
- 2) a) Consider the following molecule . Given what you have learned about hybridization theory, draw an image or images explaining the bonding situation in this molecule. I want you to draw out all of the orbitals, hybrid orbitals and how they overlap to form the bonds in the molecule. Indicate the % s or p character in the given atomic and hybrid orbitals. Which C-C bond or bonds are the longest? In a paragraph or so explain the image or images you just drew. b) Lastly, consider the molecule below. Indicate the Molecular formula, the molar mass, label the hybridization of each atom except for hydrogen, indicate any chiral centers with a *, which bond or bonds are the shortest, identify by name of each functional group with an arrow pointing to the group.arrow_forwardDescribe each highlighted bond in terms of the overlap of atomic orbitals. (If the highlighted bond is not a pi bond, select the blank option from the dropdown menu.)arrow_forwardA T bond arises from "sideways" overlap of two parallel p orbitals. The electron density lies above and below a plane containing the 2 nuclei that is perpendicular to the orbitals. п Bonding + p-orbital p-orbital atom2 atom1 n bond What atomic or hybrid orbitals make up the T bond between C and 0 in carbon dioxide, CO2 ? orbital on C + orbital on O How many o bonds does C have in CO, ? How many T bonds does C have ? Submit Answer Retry Entire Group 9 more group attempts remaining Previous Next Email Instructor Save and Exit Cengage Learning | Cengage Technical Supportarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:9780534420123
Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:Cengage Learning
INTRODUCTION TO MOLECULAR QUANTUM MECHANICS -Valence bond theory - 1; Author: AGK Chemistry;https://www.youtube.com/watch?v=U8kPBPqDIwM;License: Standard YouTube License, CC-BY