
Organic Chemistry
9th Edition
ISBN: 9781305080485
Author: John E. McMurry
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 29.4, Problem 6P
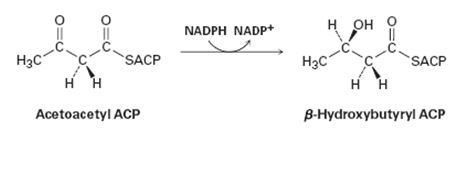
Does the reduction of acetoacetyl ACP in step 6 occur on the Re face or the Si face of the molecule?
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
which is the best method for the preparation of H2O2 in laboratory?
Which is the reductant and oxidant in the reaction?
2 KMnO4 + 5 C6H8O6 + 3 H2SO4 → 2 MnSO4 + K2SO4 + 5 C6H6O6 + 8 H2O
C6H8O6 is the reductant; KMnO4 is the oxidant
KMnO4 is the reductant; C6H8O6 is the oxidant
C6H6O6 is the reductant; MnSO4 is the oxidant
MnSO4 is the reductant; C6H6O6 is the oxidant
Chemistry
3.Calculate the amount of free energy required (or released) to allow electron transport in the ETC to be spontaneous from NADH to ubiquinone under standard conditions based on two electrons moving through the system. This occurs at 25°C.
½ O2 + 2e + 2H+ ® H2O E°´ = 0.815 V
NAD+ + 2e +H+ ® NADH E°´ = -0.315 V
NADP+ + H+ + 2e ® NADPH E°´ = -0.320 V
FAD + 2e +2H+ ® FADH2 E°´ = -0.219 V
Ubiquinone + 2H + 2e ® Ubiquinol E°´ = 0.045 V
Chapter 29 Solutions
Organic Chemistry
Ch. 29.1 - Prob. 1PCh. 29.3 - Write the equations for the remaining passages of...Ch. 29.3 - Prob. 3PCh. 29.4 - Write a mechanism for the dehydration reaction of...Ch. 29.4 - Evidence for the role of acetate in fatty-acid...Ch. 29.4 - Does the reduction of acetoacetyl ACP in step 6...Ch. 29.5 - Prob. 7PCh. 29.5 - Look at the entire glycolysis pathway, and make a...Ch. 29.6 - Prob. 9PCh. 29.7 - Prob. 10P
Ch. 29.7 - Write mechanisms for step 2 of the citric acid...Ch. 29.7 - Prob. 12PCh. 29.8 - Prob. 13PCh. 29.9 - Write all the steps in the transamination reaction...Ch. 29.9 - What -keto acid is formed on transamination of...Ch. 29.9 - Prob. 16PCh. 29.SE - Prob. 17VCCh. 29.SE - Identify the following intermediate in the citric...Ch. 29.SE - The following compound is an intermediate in the...Ch. 29.SE - Prob. 20VCCh. 29.SE - In the pentose phosphate pathway for degrading...Ch. 29.SE - Prob. 22MPCh. 29.SE - One of the steps in the pentose phosphate pathway...Ch. 29.SE - One of the steps in the pentose phosphate pathway...Ch. 29.SE - Prob. 25MPCh. 29.SE - Prob. 26MPCh. 29.SE - Prob. 27MPCh. 29.SE - Prob. 28MPCh. 29.SE - Prob. 29MPCh. 29.SE - Prob. 30MPCh. 29.SE - Prob. 31MPCh. 29.SE - Prob. 32APCh. 29.SE - Prob. 33APCh. 29.SE - Prob. 34APCh. 29.SE - Prob. 35APCh. 29.SE - Prob. 36APCh. 29.SE - Prob. 37APCh. 29.SE - Prob. 38APCh. 29.SE - Prob. 39APCh. 29.SE - Prob. 40APCh. 29.SE - Prob. 41APCh. 29.SE - Prob. 42APCh. 29.SE - Prob. 43APCh. 29.SE - Prob. 44APCh. 29.SE - Prob. 45APCh. 29.SE - Prob. 46APCh. 29.SE - Prob. 47APCh. 29.SE - Prob. 48APCh. 29.SE - Prob. 49APCh. 29.SE - Prob. 50APCh. 29.SE - In glycerol metabolism, the oxidation of...Ch. 29.SE - Prob. 52APCh. 29.SE - Prob. 53APCh. 29.SE - Prob. 54APCh. 29.SE - In step 7 of fatty-acid biosynthesis (Figure...Ch. 29.SE - Prob. 56AP
Additional Science Textbook Solutions
Find more solutions based on key concepts
Q2. Which statement best defines chemistry?
a. The science that studies solvents, drugs, and insecticides
b. Th...
Introductory Chemistry (6th Edition)
Give the IUPAC name for each compound.
Organic Chemistry
Describe the orbitals used in bonding and the bond angles in the following compounds: a. CH3O b. CO2 c. H2CO d....
Organic Chemistry (8th Edition)
Fully developed conditions are known to exist for water flowing through a 25-nim-diameer tube at 0.01 kg/s and ...
Fundamentals of Heat and Mass Transfer
45. Calculate the mass of nitrogen dissolved at room temperature in an 80.0-L home aquarium. Assume a total pre...
Chemistry: Structure and Properties
Calculate the lattice energy of CaCl2 using a Born-Haber cycle and data from Appendices F and L and Table 7.5. ...
Chemistry & Chemical Reactivity
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- From E' values in Table 14.1, calculate the equilibrium constant for the glutathione peroxidase reaction at 37 °C.arrow_forwardIllustrate the cope elimination is an intramolecular E2arrow_forward10) Can H3PO4 exist with H₂PO4 in a mixture? What about H₂PO4 and HPO4²? What about H₂PO4 and PO,³?arrow_forward
- 5. Draw the catalytic cycle for the following reaction using PCP-Ir catalyst. + + CO COHarrow_forwardExplain the two Mechanisms for the Reaction of ROH with HX ?arrow_forwardOCH3 + CH3NH2 a. Draw the structure of the tetrahedral intermediate INITIALLY-FORMED in the reaction shown. • • • You do not have to consider stereochemistry. Do not include counter-ions, e.g., Na+, I, in your answer. In cases where there is more than one answer, just draw one. √n [ ? ChemDoodle b. Draw the structures of the organic products of the acyl transfer reaction. • You do not have to consider stereochemistry. • Draw the neutral form of the products: no charges.arrow_forward
- What happens when the codeine molecule (C18H21NO3) is exposed to air? What reactions are obtained?arrow_forwardCaC2O4 + KMnO4 (in the presence of H2SO4)arrow_forwardIn PLP-catalyzed reactions, the bond broken in the substrate molecule must be perpendicular to the plane of the pyridinium ring. Considering the bonds present in this ring, describe why this arrangement stabilizes the carbanion.arrow_forward
- In Image 10 which of the following is the tetrahedral intermediate formed in the reaction of a thioester with ammonia?arrow_forwardThe dynamic process by which both forms of a- and B-D-glucopyranose in solution change slowly into an equilibrium mixture of both is known as (choose the name). OH OH Но но НО HO OH OH OH OH a-D-glucopyranose 36% B-D-glucopyranose 64% OA Acetylation O B. Enediol Rearrangement OC Mutarotation O D. Acetal Formation O E Epimerizationarrow_forwardWhat does the reaction of oxidation of Isoborneol and reduction of Camphor look like? (two different reactions total)arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you

Enzymes - Effect of cofactors on enzyme; Author: Tutorials Point (India) Ltd;https://www.youtube.com/watch?v=AkAbIwxyUs4;License: Standard YouTube License, CC-BY
Enzyme Catalysis Part-I; Author: NPTEL-NOC IITM;https://www.youtube.com/watch?v=aZE740JWZuQ;License: Standard Youtube License