
Concept explainers
(a)
Interpretation:
Nitrogen atom that is a more basic among the two nitrogen atoms present in epibatidine has to be explained.
Concept introduction:
Depending on the number of carbon side chain of the nitrogen, different types of amines can form.
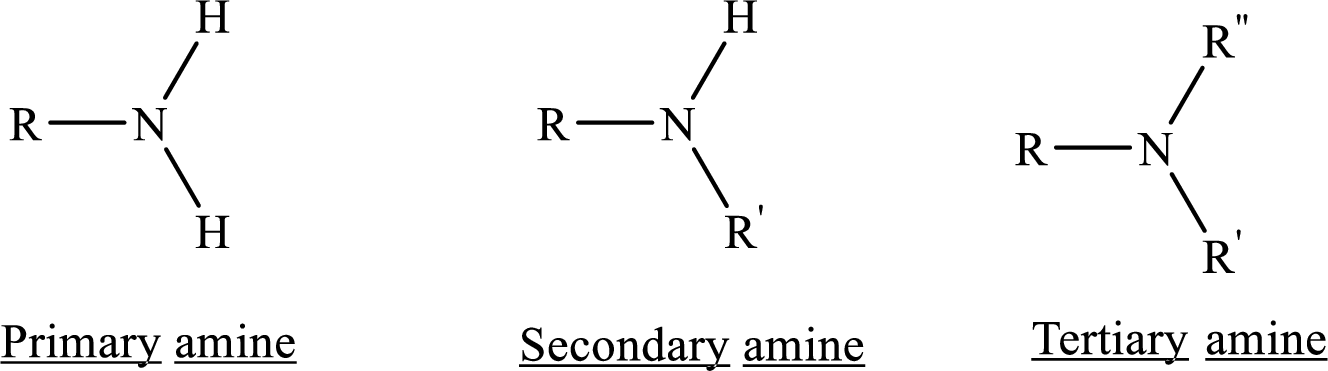
According to the explanations by Lewis, if a species donates an electron pair then it is a Lewis base whereas if a species receives an electron pair, then it is Lewis acid.
Hybridization is the mixing of valence atomic orbitals to get equivalent hybridized orbitals that having similar characteristics and energy.
(b)
Interpretation:
Chiral centers in epibatidine have to be marked.
Concept Introduction:
Amines are the derivatives of ammonia
Depending on the number of carbon side chain of the nitrogen, different types of amines can form.
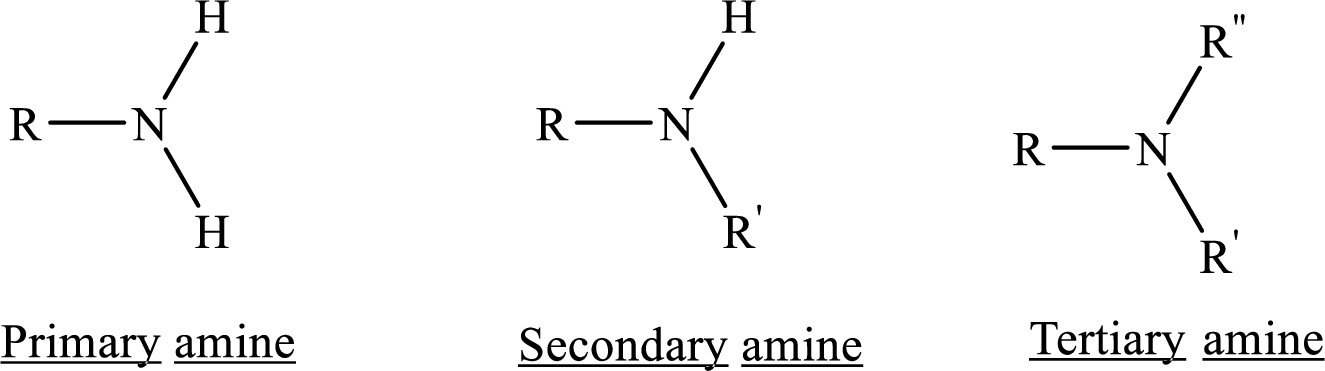
Chirality: It refers to an atom in a molecule that contains four different substituents.
Trending nowThis is a popular solution!

Chapter 23 Solutions
Organic Chemistry
- 19-33 Rank the amines in each set in order of increasing basicity. (b) sbims ne (a) NH2 NH2 N. H (c) H (d) NH2 NH2 NH2 H3C H2 (е) NH2 CH,NH, CONH,arrow_forwardGiven that C6H11COOH has a pKa = 4.8 and C6H11N+H3 has a pKa = 10.7, (a) What pH would you make the water layer to cause the carboxylic acid to dissolve in the water layer and the amine to dissolve in the ether layer? (b) What pH would you make the water layer to cause the carboxylic acid to dissolve in the ether layer and the amine to dissolve in the water layer?arrow_forwardRank the labeled N atoms in the anticancer drug imatinib (trade name Gleevec) in order of increasing basicity. Imatinib, sold as a salt with methanesulfonic acid (CH3SO3H), is used for the treatment of chronic myeloid leukemia as well as certain gastrointestinal tumors.arrow_forward
- What carboxylic acid and amine are needed to synthesize the pain reliever phenacetin? Phenacetin was once a component of the over-the-counter pain reliever APC (aspirin, phenacetin, caffeine), but it is no longer used because of its kidney toxicity.arrow_forwardDetermine which of the following bases is strong enough to deprotonate acetonitrile (CH3CN), so that equilibrium favors the products:(a) NaH; (b) Na2CO3; (c) NaOH; (d) NaNH2; (e) NaHCO3.arrow_forwardFor each of the following pairs, indicate which of the two is the stronger base: (a) F─ or I─ (b) CH3-NH2 or CH3-NH3+arrow_forward
- Provide an explanation without using the pka values : Why is phenol stronger acid than butanoic acid?arrow_forwardIn the mid-1930s a substance was isolated from a fungus that is a parasite of ryes and other grasses. This alkaloid, lysergic acid, has been of great interest to chemists because of its strange, dramatic action on the human mind. Many derivatives of lysergic acid are known, some with medicinal applications. Perhaps the best known derivative of lysergic acid is the potent hallucinogen lysergic acid diethylamide (LSD): మగవా జి N-H LSD (CH25N;O) Like other alkaloids, LSD is a weak base, with Kp = 7.6 × 107. What is the pH of a 0.94 M solution of LSD? pH =arrow_forwardParoxetine (Paxil) is an antidepressant that is a member of a family of drugs known as Selective Serotonin Reuptake Inhibitors (SSRIS). This family of drugs also includes fluoxetine (Prozac) and sertraline (Zoloft). SSRIS work by inhibiting the reuptake of the neurotransmitter serotonin in the synapses of the central nervous system follow- ing release of serotonin during excitation of individual nerve cells. Between firings, the serotonin is taken back up by a nerve cell in preparation for firing again. Inhibition of reuptake has the effect of increasing the time serotonin molecules remain in the syn- apses following excitation, leading to a therapeutic effect. In one synthesis of parox- etine, the following reagents are used. Draw the structures of synthetic intermediates A and B. F НО SOCI, A B HO Pyridinearrow_forward
- Using pKa Values to Determine Relative Acidity and Basicity Rank the following compounds in order of increasing acidity, and then rank their conjugate bases in order of increasing basicity.arrow_forwardPQ-27. What is the product of this reaction? (A) 0 OH a (B) (D) H₂ Ptarrow_forward15.44 Identify the following glycerophospholipid, which helps con- duct nerve impulses in the body, as a lecithin or cephalin, and list its components: O || CH,−O−C—(CH)4—CH, O || CH–0–C− (CH2)16—CH3 CH,−0-P-0–CH2–CH,-N–CH, O || Albo CH3 4+ CH3arrow_forward
 Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning

