
(a)
Interpretation:
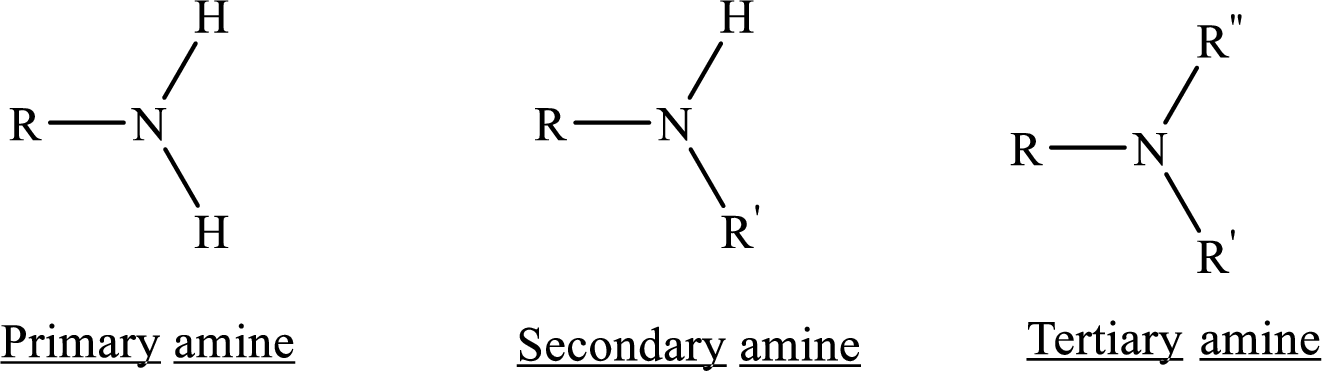
Given amine has to be classified as a primary, secondary, or tertiary amine.
Concept introduction:
In chemistry Structure is the arrangement of
Depending on the number of carbon side chain of the amide, different types of amides can form.
(b)
Interpretation:
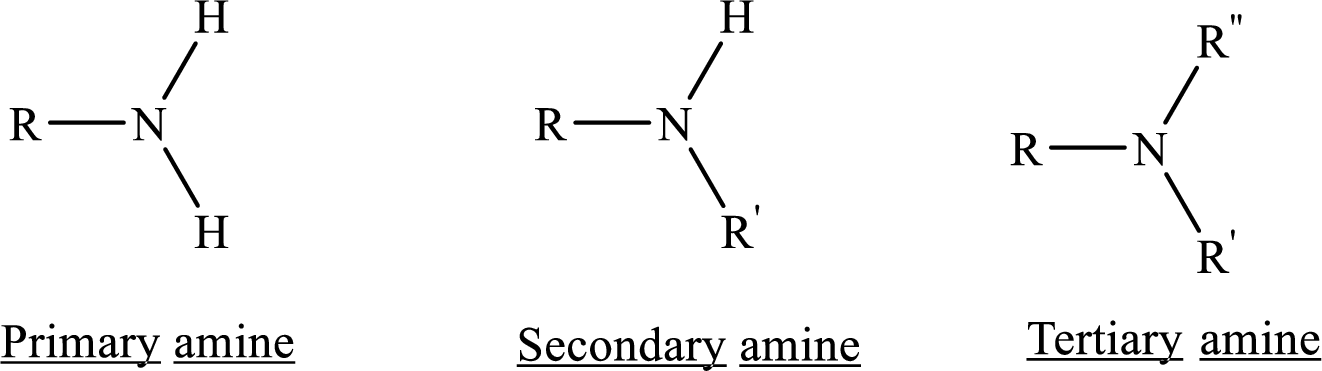
Similarities and differences between the structural formulas of (R)-epinephrine and (R)-albuterol has to be compared
Concept introduction:
In chemistry Structure is the arrangement of chemical bonds between atoms in a molecule, specifically which atoms are chemically bonded to what other atoms with what kind of chemical bond.
Amines are the derivatives of ammonia
Depending on the number of carbon side chain of the amide, different types of amides can form.
Alcohol: It is an organic compound where it contains at least one
Want to see the full answer?
Check out a sample textbook solution
Chapter 23 Solutions
Organic Chemistry
- Draw the products formed when each carbonyl compound reacts with the following amines: [1] CH3CH2CH2NH2; [2] (CH3CH2)2NH.arrow_forwardDraw the products formed when attached carbonyl compound reacts with the following amines: [1] CH3CH2CH2NH2; [2] (CH3CH2)2NH.arrow_forwardDraw the structure of a compound of molecular formula C4H11NO that ts each description: (a) a compound that contains a 1° amine and a 3° alcohol; (b) a compound that contains a 3° amine and a 1° alcohol.arrow_forward
- Draw the struetre of the following three isomeic amides with chemical feruula CHNO. Amide #1: (E)-N,N-dimethyl-2-butenamide Amide #2: (7)-N-methyl-3-pentenamide Amide #3: (7)-3-hexenamide C'onsider Z sterenchemistry of alkenes. • Draw one structure per sketcher. Add additional sketchers asing the drop-down memu in the hottom right comer. Sepamte structares with - signs from the drop-down mena. O00. IF ChemDoodle ChemDoodie ChemDoodle just the first one is what I could really use help on Amide #1: (E)-N,N-dimethyl-2-butenamidearrow_forwardDetermine the hybridization around the N atom in each amine, andexplain why cyclohexanamine is 106 times more basic than aniline.arrow_forward3 Compare the basicity of amines with other common bases, and explain how theirbasicity varies with hybridization and aromaticity.arrow_forward
- 2) Use compound C (shown below) to answer the following questions. H2N H CH3 compound C H" H3C Br a) Classify compound C as a primary, secondary or tertiary amine. b) Give the IUPAC name for compound C, omitting absolute configuration (R or S) designations. c) In the indicated spaces below, draw the enantiomer, a diastereomer and a Fischer projection of compound C. enantiomer of compound C diastereomer of compound C Fischer projection of compound Carrow_forwardRank the labeled nitrogen atoms in each compound in order of increasing basicity. Histamine causes the runny nose and watery eyes associated with allergies, and trazodone is a drug used as a sedative and antidepressant.arrow_forward1. Draw structures for the following: (a) N,N-dimethylpentanamide (b) Acetamide(c) 2,3-dimethylpentanamide (d) N-ethylbenzamidearrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

