
(a)
Interpretation:
From the given compounds from A to D, a compound containing a primary amine and a primary alcohol needs to be identified.
Concept Introduction:
Amine − is an organic N compounds formed by replacing one or more H atoms of
Alcohol- is an organic compound contains the group of
Answer to Problem 29P
Structure C.
Explanation of Solution
Amines are formed by replacing one or more hydrogen atoms of ammonia with alkyl groups.
Furthermore,
In the below stick and ball structure, C atoms are representing in black color, H atoms in white color, N atoms in blue color and O atoms in red color. Stick and ball structure can be simplified as below;
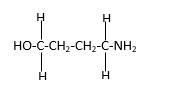
Bond that is indicated in red color is the only one
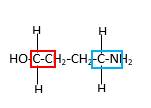
Hence, structure C is a compound that contains a
(b)
Interpretation:
From the given compounds from A to D, a compound containing a secondary amine and a secondary alcohol needs to be identified.
Concept Introduction:
Amine − is an organic N compounds formed by replacing one or more H atoms of
Alcohol- is an organic compound contains the group of
Answer to Problem 29P
Structure D.
Explanation of Solution
Amines are formed by replacing one or more hydrogen atoms of ammonia with alkyl groups.
Furthermore,
In the below stick and ball structure, C atoms are representing in black color, H atoms in white color, N atoms in blue color and O atoms in red color. Stick and ball structure can be simplified as below;
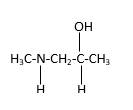
Bonds that are indicated in red color are the two
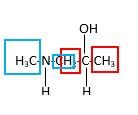
Hence, structure D is a compound that contains a
(c)
Interpretation:
From the given compounds from A to D, a compound containing a primary amine and a tertiary alcohol needs to be identified.
Concept Introduction:
Amine − is an organic N compounds formed by replacing one or more H atoms of
Alcohol- is an organic compound contains the group of
Answer to Problem 29P
Structure B.
Explanation of Solution
Amines are formed by replacing one or more hydrogen atoms of ammonia with alkyl groups.
Furthermore,
In the below stick and ball structure, C atoms are representing in black color, H atoms in white color, N atoms in blue color and O atoms in red color. Stick and ball structure can be simplified as below;
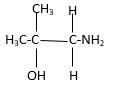
Bonds that are indicated in red color are the three
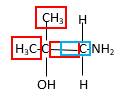
Hence, structure B is a compound that contains a
(d)
Interpretation:
From the given compounds from A to D, a compound containing a tertiary amine and a primary alcohol needs to be identified.
Concept Introduction:
Amine − is an organic N compounds formed by replacing one or more H atoms of
Alcohol- is an organic compound contains the group of
Answer to Problem 29P
Structure A.
Explanation of Solution
Amines are formed by replacing one or more hydrogen atoms of ammonia with alkyl groups.
Furthermore,
In the below stick and ball structure, C atoms are representing in black color, H atoms in white color, N atoms in blue color and O atoms in red color. Stick and ball structure can be simplified as below;
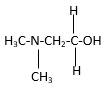
Bond that is indicated in red color is the only one
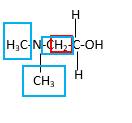
Hence, structure A is a compound that contains a
Want to see more full solutions like this?
Chapter 18 Solutions
General, Organic, and Biological Chemistry - 4th edition
- 3. Give the pharmacological property of the following alkaloids. Where do they naturally occur? a. morphine b. quinine c. berberine 4. What are the chemical tests used in identifying alkaloids?arrow_forwardClassify each of the molecules below. 1° amine 1° amine 1° amine 2° amine 2° amine 3° amine 3° amine not an amine at all not an amine at all 2° amine 3° amine not an amine at allarrow_forwardC. Amides 1. Amidation a. Acetic acid + ammoniaarrow_forward
- The hydrolysis of an amide in acidic conditions forms A. a carboxylate salt and an alcohol B. a carboxylate salt and an amine C. an alcohol and an amine salt (an ammonium ion) D. a carboxylic acid and an amine salt (an ammonium ion)arrow_forwardExplain about Simple Amines and Alkaloids ?arrow_forwarda. 1° amine b. How many nitrogen-hydrogen bonds are present in the functional group in each of the following types of amines? a. 1° amine 17-6 b. 2° amine c. 3° amine Cthe followvingarrow_forward
- Which of the following statements is INCORRECT? a. Amines are electrophilic. Amines are stronger bases than alcohols, ethers, or water. Nitrogen atom is sp³-hybridized and is tetrahedral in shape. Solubility of amines decreases with increasing number of carbons. O b. O C. O d.arrow_forwardA. 1/e/1FAlpQLSfSGcE8gf218c6JoHMWME1A8Nydf8M4g1yG93D-2LPpoMj_g/formResponse Question 24: Which compounds in Figure 24 is both a tertiary amine and a tertiary alcohol? * Compound A O Compound B O Compound C ɔ punodwoɔ a punodwoɔ O O None of these compounds fit the given description. Compound D Figure 24 HO HN N. D. OH HN OH dp 12 SUarrow_forwardCyclic amides are called which of the following? a. lactams b. lactanes c. lactones d. lactinesarrow_forward
- Erythronolide B is the biological precursor of erythromycin, a broad-spectrum antibiotic. What functional group Erythronolide B does contain? a. b. H₂CH₂C C. H₂C 1 H₂C 2 3 4 O Amide d. Amine OH Erythronolide B Ketone Aldehyde a CH₂ b C d CH₂ OH JCH₂ 'OH OHarrow_forwarda.Primary amides tend to exist as dimers in the solid and liquid state. b.Dimethylacetamide, CH3CON(CH3)2 has a higher boiling point than acetamide. c.Nitrile is often classified as an acid derivative because it is hydrolyzed to a -COOH. d.Esters have lower boiling points than ketones of comparable molecular masses. Whice are correct?arrow_forward2. What is produced when an amine reacts with a strong acid such as HCl? A. An amine and the OH- ion B. An amide and the H+ ion C. An ammonium hydroxide D. An ammonium saltarrow_forward
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,




