
(a)
Interpretation:
The acceptable name for the following ester should be determined:
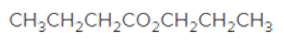
Concept introduction:
An organic compound in which carboxy
The reaction which results in the formation of at least one ester along with water on heating acids with alcohols is said to be esterification.
So, in order to give the IUPAC name to the esters, the following steps are followed:
- The alkyl substituent from the alcohol is named first.
- The name of the parent chain from carboxylic acid part is replaced as carboxylate.
(b)
Interpretation:
The acceptable name for the following ester should be determined:
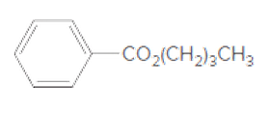
Concept introduction:
An organic compound in which carboxy functional group that is -COOH is bonded to the carbon atom is said to be a carboxylic acid. The general formula for carboxylic acid is RCOOH or RCO2H. When -H of the carboxylic acid is replaced by an alkyl or aryl group (-R') then it results in the formation of an ester having general formula RCOOR'.
The reaction which results in the formation of at least one ester along with water on heating acids with alcohols is said to be esterification.
So, in order to give the IUPAC name to the esters, the following steps are followed:
- The alkyl substituent from the alcohol is named first.
- The name of the parent chain from carboxylic acid part is replaced as carboxylate.
Want to see the full answer?
Check out a sample textbook solution
Chapter 17 Solutions
General, Organic, and Biological Chemistry - 4th edition
- 23. What reactant and catalyst is(are) needed to change ethene into ethane? a. H2, Pt b. H2O, H2SO4 c. HCI d. heat, pressurearrow_forwardWhich alkyl halide has the highest boiling point? A. CH3BrB. CH3FC. CH3ClD. CH3larrow_forwardAn example of a primary alcohol is shown by the structure CH3CH2CH(OH)CH3. A. true B. falsearrow_forward
- 1. Carboxylic acid reacts with an alcohol to form: A. Ester and Water B. Ester C. Water D. No reaction 2. The general formula for Carboxylic acids: A. RCOOH B. RCOOR C. RCOR D. RCOH 3. General formula of phenols: * A. ROH B. Ar-OH C. R-SH D. RCOHarrow_forwardDraw the structure of each carboxylic acid or ester. a. pentanoic acid b. methyl hexanoate c. 3-ethylheptanoic acid d. butyl ethanoatearrow_forwardH2SO4 5. What is the product of the following reaction? R'OH C. alcohol D. alkyne A. ester B. aldehyde H Harrow_forward
- 8 What is the organic product? A. T B. + I HI D. ? Iarrow_forwardMULTIPLE CHOICE 1. They impart fragrance of fruit and flowers. * A. alcohol B. ether C. carboxylic acid D. ester 2. They are the final oxidation product of primary alcohol. * A. ester B. aldehyde C. carboxylic acid D. ketonearrow_forward4. A student prepared an ester using a different alcohol and a different carboxylic acid than the one you used in lab: It H-C-C-C-OH H-C-C-Ċ -C-0H H H It butanoič acid H. propanol a. Draw the structural formula of the ester that formed. b. Name the esterarrow_forward
- Draw the products a. Pentyl butanoate reacts with water and NaOH b. Acetic anhydride reacts with ethanol c. 3-hexanone reacts with two equivalents of isopropyl alcohol d. Hexanal reacts with NaBH4 and CH3OHarrow_forward24 Which of the following contains a hydroxyl group? A. Alcohol B. Ether C. Thiol D. More than one answer is correct 25 Tetrahydrocannabinol molecule has four functional groups. Which one is not included? A. An alkane B. an ether C. a phenolic -OH D. benzene ring E. a carbon-carbon double bond H H3C CH3 H3C H OH CH3arrow_forwardWhat test can differentiate ethene and ethanol? a. Fehling's Test b. Tollens' Test c. KMNO4 d. K2Cr207arrow_forward
 Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning



