
Concept explainers
Interpretation:
The labeled
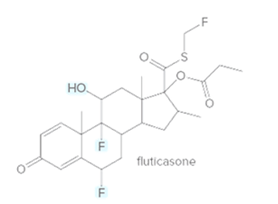
Concept Introduction:
The compounds in which hydrogen atoms of an
When the halogen is present on the carbon atom and that carbon atom is bonded to one other carbon atom is known as primary (
When the halogen is present on the carbon atom and that carbon atom is bonded to two other carbon atoms is known as secondary (
When the halogen is present on the carbon atom and that carbon atom is bonded to three other carbon atoms is known as tertiary (
When the hydroxyl group (-OH) is present on the carbon atom and that carbon atom is bonded to one other carbon atom is known as primary (
When the hydroxyl group (-OH) is present on the carbon atom and that carbon atom is bonded to two other carbon atoms is known as secondary (
When the hydroxyl group (-OH) is present on the carbon atom and that carbon atom is bonded to three other carbon atoms is known as tertiary (
Want to see the full answer?
Check out a sample textbook solution
Chapter 14 Solutions
General, Organic, and Biological Chemistry - 4th edition
- 18-18 Propanoic acid and methyl acetate are constitutional isomers, and both are liquids at room temperature. One of these compounds has a boiling point of 141°C; the other has a boiling point of 57°C. Which compound has which boiling point? Explain.arrow_forwardConsider the following two molecules and answer the following questions. Molecule A: НО C. CH-CH3 CH3 Molecule B: H;C-CH,- -C-0-CH3 a) What are all of the IMFS present in molecule A? b) What are all of the IMFS present in molecule B? c) Which would have the higher boiling point, molecule A or molecule B? d) What is the relationship between molecules A & B: structural isomers, geometric isomers, identical, or not related?arrow_forwardCodeine can treat pain and cough but has a high risk for addiction and dependence. Which of the following statements about Codeine is false? H3C H- N-CH3 HO O The methoxy group masks an important binding group O The structure is converted to morphine in the brain O The structure is a weaker analgesic than morphine. O The structure acts as a prodrugarrow_forward
- Classify each of the molecules below. x NH₂ 1° amine 2° amine 3° amine not an amine at all NH₂ 1° amine 2° amine 3° amine not an amine at all NH₂ 1° amine 2° amine 3° amine not an amine at allarrow_forwardWhy are some physicians are careless when prescribing benzodiazepines for patients suffering from severe anixety?arrow_forwardThe following amines have the same molecular formula (C5H13N), but their boiling points are significantly different. Explain why. H `NH2 2-Methylbutan-1-amine Boiling point = 97 °C N-Methylbutan-2-amine Boiling point = 84 °C N-Ethyl-N-methylethan-1-amine Boiling point = 65 °Carrow_forward
- Label each nitrogen-containing functional group in lidocaine, a local anesthetic, as an amine or amide, and classify it as 1 °, 2 °, or 3 °.arrow_forwardClassify the following amines and alcohols as primary (1°), secondary (2°), or tertiary (3°). Circle the correct classification. a) d) 1° 2° 3° 1° 2° 3° .OH NH₂ b) OH 88 1° 2° 3° OH h) 1° 2° 3° NH₂ 1° 2° 3° i) 1° 2° 3° HO ہر سڈنڈ 1° 2° 3° 1° 2° 3° 1° 2° 3°arrow_forwardThe stimulant in coffee is: a. tannic acid b. theobromine c. theophylline d. caffeinearrow_forward
- 13-27 Define autoxidation.arrow_forwardIdentify the most important aldehyde and ketone from Section 14.4 on the basis of amount used, and list at least one characteristic for each that contributes to its usefulness.arrow_forwardLabel each of the following structures as a hemiacetal, hemiketal, acetal, ketal, or none of these: a. c. b.arrow_forward
 Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,





