
Concept explainers
(a)
Interpretation:
The alcohol that is needed as a starting material to prepare the following compound as a product should be determined:
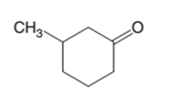
Concept Introduction:
A
In a chemical reaction, the substance which is involved in conversion is said to be reactant whereas the newly formed substance is called a product. Both reactants and products must be separated by an arrow.
The oxidation reaction is the reaction that involves the addition of O atom in the presence of certain oxidizing agents such as
(b)
Interpretation:
Interpret the alcohol that is needed as a starting material to prepare given carbonyl compound by an oxidation reaction.
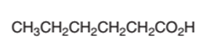
Concept Introduction:
A chemical reaction is the symbolic representation of the conversion of substances to new substances.
In a chemical reaction, the substance which is involved in conversion is said to be reactant whereas the newly formed substance is called a product. Both reactants and products must be separated by an arrow.
The oxidation reaction is the reaction that involves the addition of O atom in the presence of certain oxidizing agents such as
Want to see the full answer?
Check out a sample textbook solution
Chapter 14 Solutions
General, Organic, and Biological Chemistry - 4th edition
- Explain Structure and Bonding of Alcohols, ethers, and epoxides ?arrow_forwardWhat alcohol is needed to form the alkene as a product of a dehydration reaction?arrow_forwardSome methods of preparing aldehydes and ketones involve oxidation or reduction in which an alcohol, hydrocarbon true O False O * Griniard reagent with RCN product Aldehydes and ketones O ketones O aldehydes O phenol Oarrow_forward
- What organic compound can be produced from the oxidation of alcohols and aldehydes? alcohols carboxylic acids ethers esters What organic reaction is displayed in the picture below? * H H NaOH H C- H Ethanol heat H H Cl H- -H Alkyl halide substitution elimination oxidation addition alkenearrow_forwardWhat alcohol is formed when each carbonyl compound is treated with H 2 and a Pd catalyst?arrow_forwardName each compound as a phenol.arrow_forward
- What alcohol can be oxidized to each carboxylic acid?arrow_forwardGive the structure of an alcohol that could be used to prepare each of the following compounds: a. b. c.arrow_forwardList the following compounds in order of increasing water solubility: a.ethoxyethane b.propanoic acid c.pentane d.1 butanolarrow_forward
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning- Chemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
 Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning





