
Essential Organic Chemistry (3rd Edition)
3rd Edition
ISBN: 9780321937711
Author: Paula Yurkanis Bruice
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Question
Chapter 11, Problem 44P
Interpretation Introduction
Interpretation:
To predict the products obtained upon the hydrolysis of aspartame in an aqueous solution of hydrochloric acid.
Concept introduction:
Amides are generally very stable compounds however upon acid or base catalyzed hydrolysis the amide bond
The general mechanism for the acid hydrolysis of amides is given as,
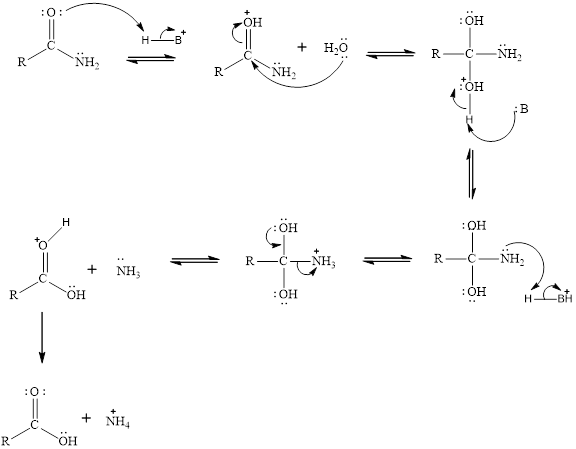
Acid: Esters undergoes acid hydrolysis the which produces alcohols.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Aspartame, the sweetener used in the commercial products NutraSweet and Equal, is 200 times sweeter than sucrose. What products will be obtained ifaspartame is hydrolyzed completely in an aqueous solution of HCl?
Why does a substance become more soluble in a solvent with increasing temperature?
Solvents are ordered by decreasing value of theirdielectric constant. In the table, which is the most polar solvent and which is the least polar?
How can acetylsalicylic acid be obtained from salicylic acid?
Aspartame, the sweetener used in the commercial products NutraSweet and Equal, is 200 times sweeter than sucrose. What products will be obtained if aspartame is hydrolyzed completely in an aqueous solution of HCl?
Chapter 11 Solutions
Essential Organic Chemistry (3rd Edition)
Ch. 11.1 - The aromas of many flowers and fruits are due to...Ch. 11.1 - Name the following compounds:Ch. 11.1 - Prob. 3PCh. 11.2 - Prob. 4PCh. 11.2 - Prob. 5PCh. 11.4 - a. What is the product of the reaction of acetyl...Ch. 11.4 - Prob. 7PCh. 11.5 - Using the pKa values listed in Table 11.1, predict...Ch. 11.6 - Starting with acetyl chloride, what neutral...Ch. 11.6 - Prob. 10P
Ch. 11.7 - Prob. 11PCh. 11.8 - Prob. 13PCh. 11.8 - Using the mechanism for the acidcatalyzed...Ch. 11.8 - Prob. 15PCh. 11.8 - Prob. 16PCh. 11.8 - Prob. 17PCh. 11.9 - Prob. 18PCh. 11.10 - Show how each of the following esters could be...Ch. 11.11 - Which of the following reactions would lead to the...Ch. 11.12 - Prob. 22PCh. 11.12 - Prob. 23PCh. 11.13 - Prob. 24PCh. 11.13 - Prob. 25PCh. 11.14 - Prob. 26PCh. 11.14 - Prob. 27PCh. 11.14 - Prob. 28PCh. 11.15 - Prob. 29PCh. 11.15 - How would you synthesize the following compounds...Ch. 11 - Write a structure for each of the following a. N,N...Ch. 11 - Prob. 32PCh. 11 - Which ester is more reactive, methyl acetate or...Ch. 11 - What products would be formed from the reaction of...Ch. 11 - What products would be obtained from the following...Ch. 11 - Prob. 36PCh. 11 - a. Which compound would you expect to have a...Ch. 11 - a. List the following esters in order of...Ch. 11 - D. N. Kursanov, a Russian chemist, proved that the...Ch. 11 - Prob. 40PCh. 11 - Using an alcohol for one method and an alkyl...Ch. 11 - Prob. 42PCh. 11 - Prob. 44PCh. 11 - Prob. 45PCh. 11 - Prob. 46PCh. 11 - Prob. 47PCh. 11 - Prob. 48PCh. 11 - Prob. 49PCh. 11 - Show how the following compounds could be prepared...Ch. 11 - Prob. 51PCh. 11 - Prob. 52PCh. 11 - Prob. 53P
Knowledge Booster
Similar questions
- Digitalis is a preparation made from the dried seeds and leaves of the purple foxglove, Digitalis purpurea, a plant native to southern and central Europe and cultivated in the United States. The preparation is a mixture of several active components, including digitalin. Digi- talis is used in medicine to increase the force of myocardial contraction and as a conduction depressant to decrease heart rate (the heart pumps more forcefully but less often). HC OH H,C H CH3 H. H. (a) Describe this glycosidic bond OCH, H A (b) Draw an open-chain Fischer projection of this monosaccharide CH3 H. (e) Describe this glycosidic bond OCH, HA H. HO H) H. OH НО (d) Name this monosaccharide unit H. OH Digitalinarrow_forwardProstaglandins are a class of eicosanoids, fatty acid derivatives with a variety of extremely potent actions on vertebrate tissues. They are responsible for producing fever and inflammation and its associated pain. Prostaglandins are derived from the 20- carbon fatty acid arachidonic acid in a reaction catalyzed by the enzyme prostaglandin endoperoxide synthase. This enzyme, a cyclooxygenase, uses oxygen to convert arachidonic acid to PGG2, the immediate precursor of many different prostaglandins. Rate of formation of PGG2 with 10 mg/ml ibuprofen (mM/min) Arachidonic acid (mM) Rate of formation of PGG2 (mM/min) 0.190 12.3 0.228 13.9 0.342 17.5 0.570 1.33 22.2 28.8 7.71 8.88 11.9 16.3 24.0 The kinetic data given in the table are for the reaction catalyzed by a mutant of prostaglandin endoperoxide synthase. Focusing here on the first two columns, determine the Vmax and Km of the enzyme. Vmax Km = mM/min mMarrow_forwardThere is a tick and mosquito repellent DEET (diethyltoluamide) that is prepared by a reaction of diethylamine with m-methylbenzomic acid (m-toluic acid) H+ and heat. What's the structure of DEET?arrow_forward
- Look up the structure of lisdexamfetamine (Vyvanse), a drug used in the treatment of attention deficit hyperactivity disorder (ADHD). Redraw it and identify all the functional groups present. What is known about itstherapeutic properties?arrow_forwardAspirin is the common name for the compound acetylsalicylic acid, widely used as a fever reducer and as a pain killer. Salicylic acid, whose name comes from Salix, the willow family of plants, was derived from willow bark extracts. In folk medicine, willow bark teas were used as headache remedies and other tonics. Nowadays, salicylic acid is administered in the form of aspirin which is less irritating to the stomach than salicylic acid. To prepare aspirin, salicylic acid is reacted with an excess of acetic anhydride. A small amount of a strong acid is used as a catalyst which speeds up the reaction. In this experiment, phosphoric acid will be used as the catalyst. The excess acetic acid will be quenched with the addition of water. The aspirin product is not very soluble in water so the aspirin product will precipitate when water is added. The synthesis reaction of aspirin is shown below: Actic anhydride 5 ml. Acetic acid Salicylic acid 28 Acetylsalicylie acid Procedure 1) Mix salicylic…arrow_forwardEthyl butyrate, CH3CH2CH2CO2CH2CH3, is an artificial fruit flavor commonly used in the food industry for such flavors as orange and pineapple. Its fragrance and taste are often associated with fresh orange juice, and thus it is most commonly used as orange flavoring.It can be produced by the reaction of butanoic acid with ethanol in the presence of an acid catalyst (H+): CH3CH2CH2CO2H(l)+CH2CH3OH(l)H+⟶CH3CH2CH2CO2CH2CH3(l)+H2O(l) a) Given 7.70 g of butanoic acid and excess ethanol, how many grams of ethyl butyrate would be synthesized, assuming a complete 100% yield? b) A chemist ran the reaction and obtained 5.25 g of ethyl butyrate. What was the percent yield? c) The chemist discovers a more efficient catalyst that can produce ethyl butyrate with a 78.0% yield. How many grams would be produced from 7.70 g of butanoic acid and excess ethanol?arrow_forward
- Ethyl butyrate, CH3CH2CH2CO2CH2CH3, is an artificial fruit flavor commonly used in the food industry for such flavors as orange and pineapple. Its fragrance and taste are often associated with fresh orange juice, and thus it is most commonly used as orange flavoring.It can be produced by the reaction of butanoic acid with ethanol in the presence of an acid catalyst (H+): CH3CH2CH2CO2H(l)+CH2CH3OH(l)H+⟶CH3CH2CH2CO2CH2CH3(l)+H2O(l). The chemist discovers a more efficient catalyst that can produce ethyl butyrate with a 78.0% yield. How many grams would be produced from 8.50 gof butanoic acid and excess ethanol? Express your answer in grams to three significant figures.arrow_forwardEthyl butyrate, CH3CH2CH2CO2CH2CH3, is an artificial fruit flavor commonly used in the food industry for such flavors as orange and pineapple. Its fragrance and taste are often associated with fresh orange juice, and thus it is most commonly used as orange flavoring.It can be produced by the reaction of butanoic acid with ethanol in the presence of an acid catalyst (H+): CH3CH2CH2CO2H(l)+CH2CH3OH(l)H+⟶CH3CH2CH2CO2CH2CH3(l)+H2O(l) Given 8.50 g of butanoic acid and excess ethanol, how many grams of ethyl butyrate would be synthesized, assuming a complete 100%yield? Express your answer in grams to three significant figures.arrow_forwardare involved in the regulation of glucose metabolism and in control tissue swelling by the control of inflammation, while regulating cellular salt balance between Na+ and K+. a.glucocorticoids, mineralocorticoids b. mineralocorticoids, glucocorticoids c. androgen, estrogen d. estrogen, androgen Which of the following represents an aldonic acid? OH OH но, но OH но —н но Но HO HO H но но H H- -OH -OH H OH H FOH H- -OH- OH HO но HO. OH II III IV V Refer to the structures. Which of the following compounds will react with acetyl chloride to give an amide and ester, respectively? NH2 ÇOOH CH3 он CHO II II IV V The six groups of enzymes are the following: I. Oxidoreductases II. Transferases III. Hydrolases IV. Isomerases V. Lyases VI. Ligases Fill in the blanks: catalyze hydrolysis while catalyze bond formation.arrow_forward
- Aspirin is an anti-inammatory agent because it inhibits the conversion of arachidonic acid to prostaglandins by the transfer of its acetyl group (CH3CO –) to an OH group at the active site of an enzyme (Section 19.6). This reaction, called transesterication, results in the conversion of one ester to another by a nucleophilic acyl substitution reaction. Draw a stepwise mechanism for the given transesterication.arrow_forwardDescribe the reaction where benzil is prepared by the oxidation of an a-hydroxyketone, benzoin and uses nitric acid to preform oxidation.arrow_forwarda. (S)-Penicillamine, an amino acid that does not occur in proteins, is used as a copper chelating agent to treat Wilson's disease, an inherited defect in copper metabolism. (R)-Penicillamine is toxic, sometimes causing blindness. Draw the structures of (R)- and (S)- penicillamine. b. What disulfide is formed from oxidation of (S)-penicillamine?arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning

Chemistry for Today: General, Organic, and Bioche...
Chemistry
ISBN:9781305960060
Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:Cengage Learning


Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:9781285869759
Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:Cengage Learning