
(a)
Interpretation:
The 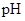 of water at given temperatures should be determined.
of water at given temperatures should be determined.
Concept Introduction:
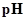 : The concentration of hydrogen ion is measured using
: The concentration of hydrogen ion is measured using 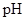 scale. The
scale. The 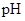 of a solution is a figure that expresses the acidity or the alkalinity of a given solution.
of a solution is a figure that expresses the acidity or the alkalinity of a given solution.
It is defined as the negative base-10 logarithm of the hydrogen or hydronium ion concentration.
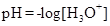
If the value of 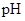 is less than
is less than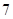 , then the solution is acidic whereas if the value of
, then the solution is acidic whereas if the value of 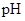 is greater than
is greater than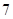 , then the solution is basic.
, then the solution is basic.
(b)
Interpretation:
The 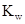 at given temperature should be determined.
at given temperature should be determined.
Concept Introduction:
Ionic-product constant for water: It is the hydronium ion concentration times the 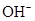 concentration present in the solution.
concentration present in the solution.
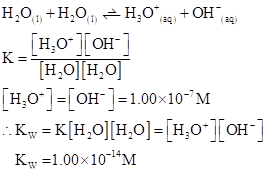
The 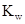 will apply to all aqueous solution.
will apply to all aqueous solution.
For acidic solution 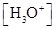 is large that is
is large that is 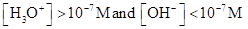
For basic solution 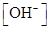 is large that is
is large that is 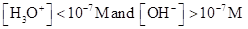
(c)
Interpretation:
The dissociation of water should be identified that whether it is an exothermic or endothermic reaction.
Concept Introduction:
Endothermic reaction: The reaction is considered as endothermic when heat energy is absorbed from surroundings during reaction.
Exothermic reaction: The reaction is considered as endothermic when heat energy is released to the surroundings during reaction.
Want to see the full answer?
Check out a sample textbook solution
Chapter 10 Solutions
Fundamentals of General, Organic, and Biological Chemistry (8th Edition)
- Calculate the pH of a solution that is 0.05000 M NH4Cl and 0.0300 M NH3 (Kb for NH3 = 1.8 x 10-5)arrow_forwardCalculate the pH of a buffer that contains 0.75 M acetic acid and 0.35 M acetate ion in 1 L solution. What will the pH of the buffer be upon the addition of 100.0 mL of 1.0 M HCI? (pKa of acetic acid 4.76)arrow_forwardConvert the following pH values in both [H+] and [OH-] values. (6 points) 1. pH = 3.95 [H+] = and [OH-] = 2. pH = 11.86 [H+] = and [OH-] = 3. pH = 4.00 [H+] = and [OH-] =arrow_forward
- A 0.200 M solution of a weak monoprotic acid (HA) has a pH of 2.35. What is the value of K of this acid? a K =2.011e-4 a What is the percent ionization of this acid? Percent Ionization = .10 %arrow_forwardBuffers are aqueous solutions that tend to resist changes in pH when small amounts of strong acid (H+) or base (OH-) are added. Compare the changes in the pH of the resulting solution when you add 0.1 M NaOH in (a) distilled water and (b) bicarbonate solution. A) (a) and (b) solutions will have the same pH. B) pH change in (b) will be higher than in (a). C) No changes in pH of both solutions. D) pH change in (a) will be higher than in (b).arrow_forwardPeople often take milk of magnesia to reduce the discomfort as- sociated with acid stomach or heartburn. The recommended dose is 1 teaspoon, which contains 4.00 x 102 mg of Mg(OH)2. What volume of an HCI solution with a pH of 1.3 can be neutralized by one dose of milk of magnesia? If the stomach contains 2.00 x 102 ml. of pH 1.3 solution, is all the acid neutralized? If not, what fraction is neutralized?arrow_forward
- Calculate the pH of a mixture of 0.25 M acetic acid and 0.20 M sodium acetate. The pKa of acetic acid is 4.76.arrow_forwardWhat is the pH of a 0.25 M solution of acetic acid, Ka = 1.8 x 105. What percentage of the acid is dissociated?arrow_forwardFor an acid HA, the concentrations of HA and A are 0.075 and 0.025, respectively, at pH 6.0. What is the p K a value for HA?arrow_forward
- Assuming equal concentrations of conjugate base and acid, which one of the following mixtures is suitable for making a buffer solution with an optimum pH of 7.4–7.6? NaOCl / HOCl (K a = 3.2 × 10 –8) NH 3 / NH 4Cl (K a = 5.6 × 10 –10) NaNO 2 / HNO 2 (K a = 4.5 × 10 –4) NaCl / HCl CH 3COONa / CH 3COOH (K a = 1.8 × 10 –5)arrow_forward200 microliters of a standard solution of 0.200 mg/mL caffeine was mixed with 4.8 mL of 50 mM sodium phosphate buffer, pH 3: methanol (60:40 v/v). What is the concentration of caffeine, in mg/mL, in this solution? Give your answer to two significant figures.arrow_forwardIf 0.752 moles of (NH3OH)Cl is dissolved in 1 L of water what is the pH of the solution?arrow_forward
 BiochemistryBiochemistryISBN:9781319114671Author:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.Publisher:W. H. Freeman
BiochemistryBiochemistryISBN:9781319114671Author:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.Publisher:W. H. Freeman Lehninger Principles of BiochemistryBiochemistryISBN:9781464126116Author:David L. Nelson, Michael M. CoxPublisher:W. H. Freeman
Lehninger Principles of BiochemistryBiochemistryISBN:9781464126116Author:David L. Nelson, Michael M. CoxPublisher:W. H. Freeman Fundamentals of Biochemistry: Life at the Molecul...BiochemistryISBN:9781118918401Author:Donald Voet, Judith G. Voet, Charlotte W. PrattPublisher:WILEY
Fundamentals of Biochemistry: Life at the Molecul...BiochemistryISBN:9781118918401Author:Donald Voet, Judith G. Voet, Charlotte W. PrattPublisher:WILEY BiochemistryBiochemistryISBN:9781305961135Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougalPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305961135Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougalPublisher:Cengage Learning BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning Fundamentals of General, Organic, and Biological ...BiochemistryISBN:9780134015187Author:John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. PetersonPublisher:PEARSON
Fundamentals of General, Organic, and Biological ...BiochemistryISBN:9780134015187Author:John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. PetersonPublisher:PEARSON





