
Concept explainers
(a)
Interpretation:
Lewis structure for
Concept Introduction:
Lewis structure is used for predicting the shape of molecules. From the steric number obtained in a Lewis structure, the molecular geometry can be predicted. VSEPR model can predict the shape of molecules considering their Lewis structure. Certain rules has to be followed in for the VSEPR model.
- The molecule will have a shape where there is minimal electrostatic repulsion between the valence‑shell electron pairs.
- The forces of repulsion between two lone pairs of electrons will be higher than the repulsion between lone pair and bond pair of electrons. This in turn will be higher than the bond pair‑bond pair of electrons.
(a)
Explanation of Solution
Lewis structure for
The total number of valence electrons present in
The skeletal structure can be drawn considering the formula as shown below;
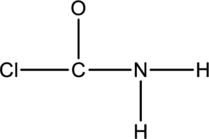
Ten electrons are used up in the skeletal structure. Among the remaining fourteen valence-electrons, six are placed over chlorine atom and six are placed over oxygen atom. Two electrons are placed over nitrogen atom.
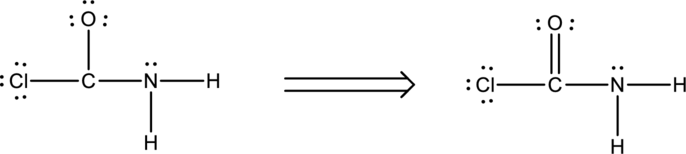
Thus the Lewis structure of
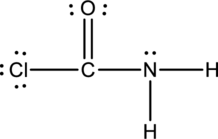
Bond Angles:
There is a carbon atom and a nitrogen atom as central atom in the Lewis structure. The steric number of the central atom can be used to predict the bond angles of the central atoms.
Steric number for carbon
The number of lone pair of electrons on carbon atom is zero while the number of atoms that are bonded to carbon is three. Therefore, steric number can be calculated as shown below;
As the steric number is three, and only bonding pair of electrons are present, the arrangement is trigonal planar and the bond angle will be
Steric number for nitorgen atom:
The number of lone pair of electrons on nitrogen atom is one while the number of atoms that are bonded to nitrogen is three. Therefore, steric number can be calculated as shown below;
As the steric number is four, the arrangement is tetrahedral and the bond angle will be
(b)
Interpretation:
Lewis structure for
Concept Introduction:
Refer part (a).
(b)
Explanation of Solution
Lewis structure for
The total number of valence electrons present in
The skeletal structure can be drawn considering the formula as shown below;
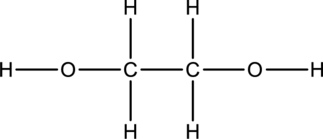
Eighteen electrons are used up in the skeletal structure. Among the remaining eight valence-electrons, four are placed over oxygen atom each. Thus the Lewis structure for
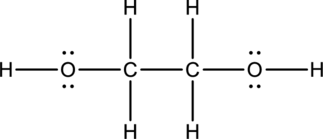
Bond Angles:
There are two carbon atoms as central atoms in the Lewis structure. The steric number of the central atom can be used to predict the bond angles of the central atoms.
Steric number for carbon atom:
The number of lone pair of electrons on carbon atom is zero while the number of atoms that are bonded to carbon is four. Therefore, steric number can be calculated as shown below;
As the steric number is four, the arrangement is tetrahedral and the bond angle will be
(c)
Interpretation:
Lewis structure for
Concept Introduction:
Refer part (a).
(c)
Explanation of Solution
Lewis structure for
The total number of valence electrons present in
The skeletal structure can be drawn considering the formula as shown below;
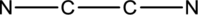
Six electrons are used up in the skeletal structure. Among the remaining twelve valence-electrons, six are placed over nitrogen atom each. Thus the Lewis structure for

Bond Angles:
There are two carbon atoms as central atoms in the Lewis structure. The steric number of the central atom can be used to predict the bond angles of the central atoms.
Steric number for carbon atom:
The number of lone pair of electrons on carbon atom is zero while the number of atoms that are bonded to carbon is two. Therefore, steric number can be calculated as shown below;
As the steric number is two, the arrangement is linear and the bond angle will be
Want to see more full solutions like this?
Chapter 10 Solutions
Chemistry: Principles and Practice
- The structural formulas for ethanol, CH3CH2OH, and propene, CH;CH=CH,2, are нн H Н—С—С—0—н H-C-C=C-H нн H H H Ethanol Propene (a) Complete the Lewis structure for each molecule showing all valence electrons. (b) Using the VSEPR model, predict all bond angles in each molecule.arrow_forward(a) Triazine, C3 H3 N3, is like benzene except that in triazineevery other C¬H group is replaced by a nitrogen atom.Draw the Lewis structure(s) for the triazine molecule. (b) Estimatethe carbon–nitrogen bond distances in the ring.arrow_forwardAcetylene 1C2H22 and nitrogen 1N22 both contain a triplebond, but they differ greatly in their chemical properties.(a) Write the Lewis structures for the two substances. (b) Byreferring to Appendix C, look up the enthalpies of formationof acetylene and nitrogen. Which compound is more stable?(c) Write balanced chemical equations for the completeoxidation of N2 to form N2O51g2 and of acetylene to formCO21g2 and H2O1g2. (d) Calculate the enthalpy of oxidationper mole for N2 and for C2H2 (the enthalpy of formationof N2O51g2 is 11.30 kJ>mol). (e) Both N2 and C2H2 possesstriple bonds with quite high bond enthalpies (Table 8.3).Calculate the enthalpy of hydrogenation per mole for bothcompounds: acetylene plus H2 to make methane, CH4;nitrogen plus H2 to make ammonia, NH3.arrow_forward
- The Lewis structure of H2CS (a) Is the molecule polar or nonpolar? (b) What is the hybridization of the carbon atom? (c) What is the geometric shape of the molecule?arrow_forwardWrite Lewis structures for these compounds. Show all valence electrons. None of them contains a ring of atoms. (a) Hydrogen peroxide, H2O2 (b) Hydrazine, N2H4 (c) Methanol, CH3OHarrow_forwardNitrogen trifluoride (NF3) is used in the electronics industry to clean surfaces. NF3 is also a potent greenhouse gas. (A) Draw the Lewis structure of NF3 and determine its molecular geometry. (B) BF3 and NF3 both have three covalently bonded fluorine atoms around a central atom. Do they have the same dipole moment? (C) Could BF3 also behave as a greenhouse gas? Explain why or why not.arrow_forward
- 1. Draw the Lewis structures for each of the following ions or molecules. For each, give (i) the molecular shape, (ii) the electron pair geometry at the central atom, and (iii) the hybridization of the central atom. (a) POF3 (b) XeO₂F3+ (c) BrCl₂ (d) N3 (the central atom is N; two other N's are bonded to it) (e) PF3arrow_forwardPropylene, C3H6, is a gas that is used to form the importantpolymer called polypropylene. Its Lewis structure is given. (a) What is the total number of valence electrons in the propylenemolecule? (b) How many valence electrons are usedto make σ bonds in the molecule? (c) How many valenceelectrons are used to make π bonds in the molecule? (d) Howmany valence electrons remain in nonbonding pairs in themolecule? (e) What is the hybridization at each carbon atomin the molecule?arrow_forwarda) Methane 1CH42 and the perchlorate ion 1ClO4- 2 are both described as tetrahedral. What does this indicate about their bond angles? (b) The NH3 molecule is trigonal pyramidal, while BF3 is trigonal planar. Which of these molecules is flat?arrow_forward
- Draw the shapes of the following molecules and ions in 3-dimension. Show clearly any lone pairs of electrons on the central atom, state the number of bond pairs and lone pairs of electrons on the central atom and name the shape of the molecule or ion. (a) SiCI4, silicon tetrachloride (b) PBr3, phosphorus tribromide (c) CI2O, dichlorine oxide Provide everything stated in the instructions for each compound.arrow_forwardFor each of the following molecules or molecular ions, give the steric number, sketch and name the approximate molecular geometry, and describe the direction of any distortions from the approximate geometry due to lone pairs. In each case, the central atom is listed first and the other atoms are all bonded directly to it. (a) TeH2 (c) PCI (b) AsF3 (d) XeFarrow_forwardOxalic acid, H2C2O4, a poisonous colorless solid, is found in some vegetables such as spinach and rhubarb. It is present in concentrations well below the toxic limit, so you can't use this as a reason to refuse a helping of spinach. The order of atoms in a molecule of oxalic acid is HO2CCO2H. (a) How many unshared pairs of electrons are on each of the carbon atoms? (b) How many unshared pairs of electrons are on each of the oxygen atoms?arrow_forward
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
