
Concept explainers
Assign formal charges to each
a. 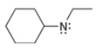 b.
b. 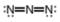 c.
c. 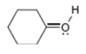 d.
d. 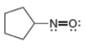
(a)
Interpretation: The formal charge to each
Concept introduction:
The formal charge on an atom is calculated by the formula,
Answer to Problem 1.40P
In the given molecule, the formal charge on nitrogen atom is
Explanation of Solution
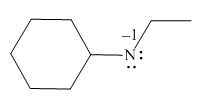
The given molecule is,
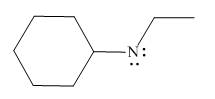
Figure 1
The formal charge on an atom is calculated by the formula,
For the nitrogen atom,
Substitute these values in above equation to calculate the formal charge on nitrogen atom.
Thus, in the given molecule, the formal charge on nitrogen atom is

Figure 2
In the given molecule, the formal charge on nitrogen atom is
(b)
Interpretation: The formal charge to each
Concept introduction: The formal charge on an atom is calculated by the formula,
Answer to Problem 1.40P
The formal charge to each
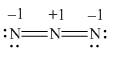
Explanation of Solution
The given molecule is,
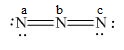
Figure 3
Here, a, b, and c are used to indicate nitrogen atoms.
The formal charge on an atom is calculated by the formula,
For the nitrogen atom,
Substitute these values in the above equation to calculate the formal charge on
In the given molecule, bond pairs and lone pairs in
Thus, the formal charge on
For the nitrogen atom,
Substitute these values in the above equation to calculate the formal charge on
Thus, the formal charge on
Hence, the formal charge to each
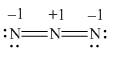
Figure 4
In the given molecule, the formal charge on nitrogen atom of both
(c)
Interpretation: The formal charge to each
Concept introduction: The formal charge on an atom is calculated by the formula,
Answer to Problem 1.40P
In the given molecule, the formal charge on oxygen atom is
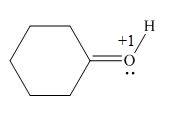
Explanation of Solution
The given species is,
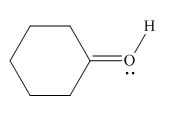
Figure 5
The formal charge on an atom is calculated by the formula,
For the oxygen atom,
Substitute these values in above equation, to calculate the formal charge on oxygen atom.
Thus, in the given molecule, the formal charge on oxygen atom is

Figure 6
In the given molecule, the formal charge on oxygen atom is
(d)
Interpretation: The formal charge to each
Concept introduction: The formal charge on an atom is calculated by the formula,
Answer to Problem 1.40P
In the given molecule, the formal charge on both nitrogen and oxygen atom is zero as shown below.
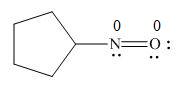
Explanation of Solution
The given species is,
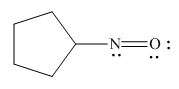
Figure 7
The formal charge on an atom is calculated by the formula,
For the oxygen atom,
Substitute these values in above equation, to calculate the formal charge on oxygen atom.
For the nitrogen atom,
Substitute these values in above equation, to calculate the formal charge on nitrogen atom.
Thus, in the given molecule, the formal charge on both nitrogen and oxygen is zero as shown below.

Figure 8
In the given molecule, the formal charge on both nitrogen and oxygen is zero.
Want to see more full solutions like this?
Chapter 1 Solutions
Organic Chemistry
Additional Science Textbook Solutions
General Chemistry: Atoms First
Organic Chemistry
Chemistry In Context
Chemistry: Structure and Properties (2nd Edition)
General, Organic, and Biological Chemistry: Structures of Life (5th Edition)
- What is the most polar bond in the molecule?arrow_forwardA. CHF i. Best Lewis Structure B. HNO (H is connected to one of the O's) i. Best Lewis Structure ii. Electron geometry on the C atom ii. Electron geometry on the N atom iii. Approximate bond angles about the C atom iii. Approximate bond angles around the N atom v. Draw the shape with in and out wedges (as necessary) and dipole arrows around the C atom. v. Draw the shape with in and out wedges (as necessary) and dipole arrows around the N atom. vi. Is the molecule polar or nonpolar? vi. Is the molecule polar or nonpolar?arrow_forwardDraw in all the hydrogen atoms and nonbonded electron pairs in each ion. a. b. d. ENHarrow_forward
- d. H E Assign formal charges to each carbon atom in the given species. All lone pairs have been drawn in. H H a. CH2-CH b. H-C-H c. H-C-H ннarrow_forwardWhich bond in each pair is more polar—that is, has the larger electronegativity difference between atoms? a. C- O or C-N b. C-F or C-Cl c. Si-C or P-Harrow_forwardDraw a Lewis structure for BF3 that obeys the octet rule if possible and answer the following questions based on your drawing. 1. For the central boron atom: - The number of lone pairs = ? - The number of single bonds = ? - The number of double bonds = ? 2. The central boron atom is a. obeys the octet rule. b. has an incomplete octet. c. has an expanded octet.arrow_forward
- To answer the questions, interpret the following Lewis diagram for SO42- . 1. For the central sulfur atom: ... The number of non-bonding electrons = The number of bonding electrons = The total number of electrons = 2. The central sulfur atom fill in the blank 4 ... A. obeys the octet rule. B. has more than an octet. C. has less than an octet.arrow_forwardCarbon ring structures are common in organic chemistry. Draw a Lewis structure for each carbon ring structure, including any necessary resonance structures. a. CHg b. CH4 c. CH12 d. C,H.arrow_forwardDraw a resonance structure that places a pi bond in a different position. Include all lone pairs in your structure. :O: H. H. 工 :O:arrow_forward
- 9. Indicate whether each statement below is true or false. a. The core electrons are called valence electrons. b. lonic bonds are formed by the attraction between cations and anions. c. Ionic bonds are formed from atoms by a transfer of electrons. d. In a Lewis symbol, the chemical symbol represents the nucleus of the atom. e. An ion written as a Lewis symbol has brackets outside the electrons. f. Two electrons involved in a bond produce a double bond. g. If the AEN value is very large the bond is polar covalent. h. The central atom is typically the atom with the highest electronegativity. i. An expanded octet has larger electron clouds. j. A nonpolar molecule can have polar bonds.arrow_forwardDraw the Lewis structure for bromoethane (C,H,Br). Be certain you include any lone pairs. C. C.arrow_forwardDetermine if a bond between each pair of atoms would be pure covalent, polar covalent, or ionic.a. Br and Br b. C and Clc. C and S d. Sr and Oarrow_forward
 Living By Chemistry: First Edition TextbookChemistryISBN:9781559539418Author:Angelica StacyPublisher:MAC HIGHER
Living By Chemistry: First Edition TextbookChemistryISBN:9781559539418Author:Angelica StacyPublisher:MAC HIGHER Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
 Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning




