
Organic Chemistry As a Second Language: Second Semester Topics
4th Edition
ISBN: 9781119110651
Author: David R. Klein
Publisher: WILEY
expand_more
expand_more
format_list_bulleted
Question
Chapter 9.3, Problem 9.19P
Interpretation Introduction
Interpretation:
Efficient synthesis for the given transformation has to be suggested.

Concept Introduction:
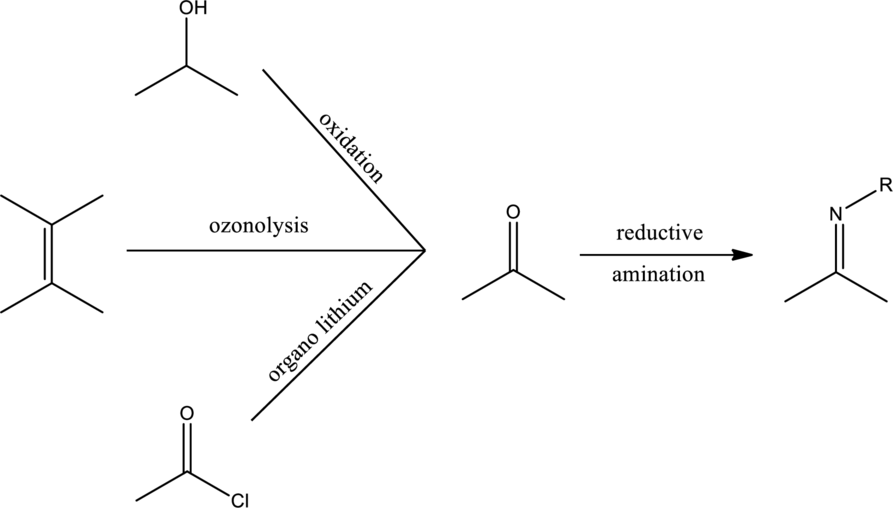
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Synthesis:
Show how you would carry out the following synthesis. Include the reagents you would need for each step and the intermediate products formed in each step. You may use any inorganic reagents you need and organic reagents with 6 or fewer carbons.
Show what reagents are necessary to make the following acetal from a ketone. Themechanism is not necessary.
Propose an efficient synthesis for each of the following transformations. Show all intermediate products, reagents and conditions.
Chapter 9 Solutions
Organic Chemistry As a Second Language: Second Semester Topics
Ch. 9.2 - Identify how you would use a Gabriel synthesis to...Ch. 9.2 - Identify how you would use a Gabriel synthesis to...Ch. 9.2 - Identify how you would use a Gabriel synthesis to...Ch. 9.2 - Identify how you would use a Gabriel synthesis to...Ch. 9.2 - Identify whether each of the following compounds...Ch. 9.2 - Identify whether each of the following compounds...Ch. 9.2 - Identify whether each of the following compounds...Ch. 9.2 - Identify whether each of the following compounds...Ch. 9.3 - Suggest an efficient synthesis for each of the...Ch. 9.3 - Suggest an efficient synthesis for each of the...
Ch. 9.3 - Suggest an efficient synthesis for each of the...Ch. 9.3 - Suggest an efficient synthesis for each of the...Ch. 9.3 - Suggest an efficient synthesis for each of the...Ch. 9.3 - Prob. 9.18PCh. 9.3 - Prob. 9.19PCh. 9.3 - Prob. 9.20PCh. 9.3 - Prob. 9.21PCh. 9.3 - Prob. 9.22PCh. 9.4 - Prob. 9.24PCh. 9.4 - Prob. 9.25PCh. 9.5 - Predict the major product for each of the...Ch. 9.5 - Predict the major product for each of the...Ch. 9.5 - Predict the major product for each of the...Ch. 9.5 - Predict the major product for each of the...Ch. 9.6 - What reagents would you use to achieve each of the...Ch. 9.6 - What reagents would you use to achieve each of the...Ch. 9.6 - What reagents would you use to achieve each of the...Ch. 9.6 - What reagents would you use to achieve each of the...Ch. 9.6 - What reagents would you use to achieve each of the...
Knowledge Booster
Similar questions
- For the following reaction schemes, fill in the appropriate reagents and starting materials you would use to transform the acetoacetic ester and acetone into nabumetone, an NSAID drug used for arthritis and joint pain. Write the correct reagents/reactants next to the number of steps for each transformation.arrow_forwardSynthesis: Show how you would carry out the following synthesis. Include the reagents you would need for each step and the structure of the intermediate products formed in each step. You may use any inorganic reagents you need and organic reagents of four or fewer carbons.arrow_forwardPropose a synthetic route to the following molecule. Use any saturated ketone, aldehyde, or ester you need (ie: you can’t use any dicarbonyl or a,b-unsaturated carbonyl compounds. You have to make those). Please show a reaction sequence.arrow_forward
- How would you carry out the following transformation? Tell what reagents you would use in each case.arrow_forwardUsing any needed reagents, show how the following synthetic transformation can be efficiently conducted.arrow_forwardBenzyl acetate is a pleasant-smelling ester found in the essential oil of jasmine flowers and is used in many perfume formulations. Starting with benzene and using any other reagents of your choice, design an efficient synthesis for benzyl acetate. The transformation above can be performed with some reagent or combination of the reagents listed below. Give the necessary reagents in the correct order, as a string of letters (without spaces or punctuation, such as "EBF"). If there is more than one correct solution, provide just one answer. A. 1) D. i ; 2) H3O + AICI 3 CI G. NBS, heat B. 1) 요. 'H H ; 2) H3O+ Ei E. H. 1) LiAlH4; 2) H3O+ CI + pyridine C. 1) F. Mg i I. Br2, AlBr3 2) H3O+arrow_forward
- Propose a multi-step synthesis of the target molecule shown at the right, using the starting materials on the left and any other reagents you need. Show the reagents needed for each step and the product of each step. You will need 4 reaction arrows.arrow_forwardHow do you write out the mechanism for this reaction? not sure what reagents to usearrow_forwardProvide an efficient synthesis for each of the following transformations. Include your retrosynthetic analysis, showing products for each step of the reaction sequence. You are not required to write a mechanism for any of these.arrow_forward
- A girl must convert 1-butyne into 2-pentanione. Assist her by recommending a synthetic plan to follow in order to obtain the desired result. Indicate all of the reagents she'll require.arrow_forwardProvide the reagents necessary to achieve the following synthetic transformations. More than one step may be required. Draw out the mechanism of the steps.arrow_forwardSelect the best reagents for each of the following transformations. You should only use each set of reagents once.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY