
Concept explainers
Answers to all problems are at the end of this book. Detailed solutions are available in the Student Solutions Manual, Study Guide, and Problems Book.
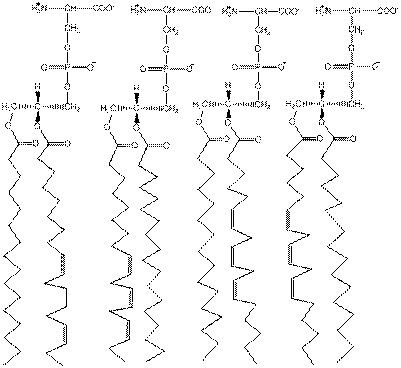
Understanding the Occurrence of Natural Phospholipids In problem 1 (b) ill Chapter 8, you weft asked to draw all the possible phosphate-dylserme isomers that form be formed from palmitic and linolenie acids. Which of the PS isomers aft not likely ll> be found in biological membranes?
Interpretation:
The PS isomer which is not likely to be found in biological membranes should be determined.
Concept Introduction:
There are two wide kinds of isomers: stereoisomers and constitutional.
Constitutional isomers vary in connectivity and bonding, whereas stereoisomers vary in 3-D orientation.
Answer to Problem 1P
Two structures having linolenic acid is connected to the initial carbon and palmitic acid is attached to the second carbon.
Explanation of Solution
Lipid is in proximity within biological membranes. Formation of double bond results in the bend in the chain which creates space among lipids.
Phosphatidyl serine molecules contain the big group on glycerol’s third carbon. Due to this group, the 2nd and 3rd carbons of glycerol contain more space as compared to the first carbon.
The fatty acid that attaches to the first carbon is usually a saturated fatty acid. An unsaturated fatty acid attaches to the second carbon.
Phosphatidylserines contain the phosphate bond in the serine residue on the 3rd carbon of glycerol.
Linoleic acid contains two common forms, thus, there are four kinds of different phosphatidylserines created with linoleic and palmitic acid.
The structure is shown as follows:
Want to see more full solutions like this?
Chapter 9 Solutions
BIOCHEM. II-EBOOK ACCESS>CUSTOM<
- Answers to all problems are at the end of this book. Detailed solutions are available in the Student Solutions Manual, Study Guide, and Problems Book. (Historical Context) The Third Person of the -Helix Publication Who was Herman Branson? What was his role in the elucidation of the structure of the or-helix'.' Did he receive sufficient credit and recognition for his contributions? And how did the rest of his career unfold? Do a Google search on Herman Branson to learn about his life, and read the article by David Eisenberg under Further Reading. You may also wish to examine the original paper by Pauling, Corey, and Branson, as well as the following Web site: http://www.pirns. org/sitelmisclclassicsl..shtml Pauling, L., Corey, R. B., and Branson, H. R., 1951. The structure of proteins: two hydrogen-bonded helical configurations of the polypeptide chain. Proceedings of the National Academy of Sciences, USA 37:235-240.arrow_forwardAnswers to all problems are at the end of this book. Detailed solutions are available in the Student Solutions Manual, Study Guide, and Problems Book. Exploring the Dimensions of the α-Helix and Coiled Coils Imagine that the dimensions of the alpha helix were such that there were exactly 3.5 amino acids per turn instead of 3.6. What would be the consequences for coiled-coil structures?arrow_forwardAnswers to all problems are at the end of this book. Detailed solutions are available in the Student Solutions Manual, Study Guide, and Problems Book. Draw all the possible isomers of threonine and assign (R,S) nomenclature to each.arrow_forward
- Answers to all problems are at the end of this book. Detailed solutions are available in the Student Solutions Manual, Study Guide, and Problems Book. Drawing Haworth Structures of Sugars Draw Haworth structures for the two possible isomers of D-altrose (Figure 7.2) and D-psicose (Figure 7.3).arrow_forwardAnswers to all problems are at the end of this book. Detailed solutions are available in the Student Solutions Manual, Study Guide, and Problems Book. Assessing the pH Dependence of Poly-L-Glutamate Structure Poly-L glutamate adopts an tr-helical structure at low pH but becomes a random coil above pH 5. Explain this behavior.arrow_forwardAnswers to all problems are at the end of this book. Detailed solutions are available in the Student Solutions Manual, Study Guide, and Problems Book. Determining the Systematic Name for a Trisaccharide Give the systematic name for it stachyose.arrow_forward
- Answers to all problems are at the end of this book. Detailed solutions are available in the Student Solutions Manual, Study Guide, and Problems Book. The dissociation constant for a particular protein dimer is 1 micromolar. Calculate the free energy difference for the monomer-to-dimer transition.arrow_forwardAnswers to all problems are at the end of this book. Detailed solutions are available in the Student Solutions Manual, Study Guide, and Problems Book. Drawing Fischer Projection Formulas for Amino Acids Without consulting chapter figures, draw Fischer projection formulas for glycine, aspartate, leucine, isoleucine, methionine, and threonine.arrow_forwardAnswers to all problems are at the end of this book.. Detailed solutions are available in the Student Solutions Manual, Study Guide, and Problems Book. Naming Sugars in the (R,S) System Use the inform tit ion in the Critical Developments in Biochemistry box titled Rules for Description (if Chiral Centers in the (R.S) System (Chapter 4) to name D-galactose using (R.S) nomenclature. Do the same for L-altrone.arrow_forward
- Answers to all problems are at the end of this book. Detailed solutions are available in the Student Solutions Manual, Study Guide, and Problems Book. Writing Dissociation Equations for Amino Acids Write equations fur the ionic dissociations of alanine, glutamate, histidine, lysine, and phenylalanine.arrow_forwardAnswers to all problems are at the end of this book. Detailed solutions are available in the Student Solutions Manual, Study Guide, and Problems Book. (Research Problem) The Nature and Roles of Linear Motifs in Proteins In addition to domains and modules, there are other significant sequence patterns in proteins—known as linear motifs—that are associated with a particular function. Consult the biochemical literature to answer the following questions: 1. What are linear motifs? 2. How are they different from domains?. 3. What are their functions? 4. How can they be characterized? 5. There are several papers that are good starting points for this problem. Neduva, V., and Russell, R., 2005. Linear motifs: evolutionary interaction switches. FEBS Letters 579:3342-3345. Gibson, T., 2009. Cell regulation: determined to signal discrete cooperation. Trends in Biochemical Sciences 34:471-482. Diella, K. Haslam, N., Chica., C. et aL, 2009. Understanding eukaryotic linear motifs and their role in cell signaling and regulation. Frontiers of Bioscience 13:6580-6603.arrow_forwardAnswers to all problems are at the end of this book. Detailed solutions are available in the Student Solutions Manual, Study Guide, and Problems Book. Use examples from the ActiveModel for Human GaleLtin-1 to describe the hydrophobic effect.arrow_forward
 BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning
