
(a)
Interpretation: All constitutional isomers formed in the given
Concept introduction: The removal of halide and neighboring
Answer to Problem 8.33P
All constitutional isomers formed in the given
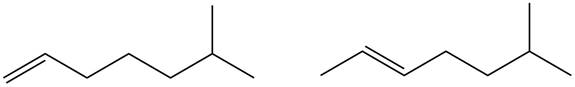
Figure 1
Disubstituted alkene is the major product of the reaction.
Explanation of Solution
The
In the given compound, two
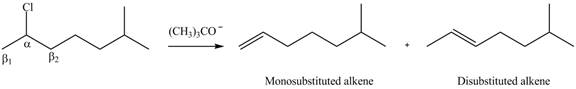
Figure 2
According to Zaitsev rule, more substituted alkene is obtained as a major product in
All constitutional isomers formed in the given
(b)
Interpretation: All constitutional isomers formed in the given
Concept introduction: The removal of halide and neighboring
Answer to Problem 8.33P
All constitutional isomers formed in the given
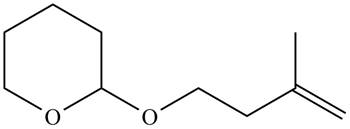
Figure 3
Monosubstituted alkene is the major product of the reaction.
Explanation of Solution
The
In the given compound, only one
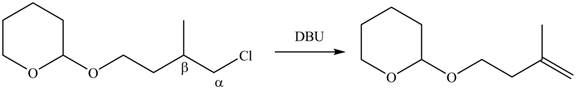
Figure 4
According to Zaitsev rule, more substituted alkene is obtained as a major product in
All constitutional isomers formed in the given
(c)
Interpretation: All constitutional isomers formed in the given
Concept introduction: The removal of halide and neighboring
Answer to Problem 8.33P
All constitutional isomers formed in the given
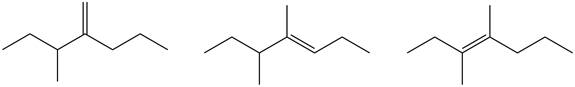
Figure 5
Tetrasubstituted alkene is the major product of the reaction.
Explanation of Solution
The
In the given compound, three
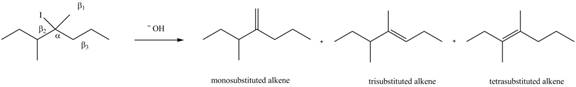
Figure 6
According to Zaitsev rule, more substituted alkene is obtained as a major product in
All constitutional isomers formed in the given
(d)
Interpretation: All constitutional isomers formed in the given
Concept introduction: The removal of halide and neighboring
Answer to Problem 8.33P
All constitutional isomers formed in the given
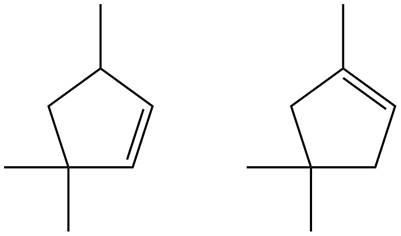
Figure 7
Trisubstituted alkene is the major product of the reaction.
Explanation of Solution
The
In the given compound, two
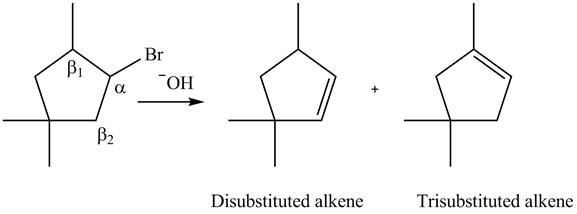
Figure 8
According to Zaitsev rule, more substituted alkene is obtained as a major product in
All constitutional isomers formed in the given
Want to see more full solutions like this?
Chapter 8 Solutions
Organic Chemistry
- Electrophilic Addition Soubong neblA-aleid rose 9160910 31 babeen ene singo 14.43 Draw the products formed when each compound is treated with one equivalent of HBr. a. b. C.arrow_forwardSynthesize each compound from acetylene. You may use any other organic or inorganic reagents. e. CH₂CH₂CH₂CCl₂CH3 f. CH₂CH₂CH₂CH₂CH₂CH₂CH3arrow_forwardDraw all constitutional isomers formed in attached E2 reaction, and predictthe major product using the Zaitsev rule.arrow_forward
- Sulfur ylides, like the phosphorus ylides, are usefulintermediates in organic synthesis. Methyl trans-chrysanthemate, anintermediate in the synthesis of the insecticide pyrethrin I,can be prepared from diene A and a sulfur ylide. Draw a stepwisemechanism for this reaction.arrow_forwardDraw the products of radical chlorination and bromination of each compound. For which compounds is a single constitutional isomer formed for both reactions? What must be true about the structure of a reactant for both reactions to form a single product? a. b. C. d. е.arrow_forwardWhich undergoes electrophilic substitution on the ring most rapidly? a. b. CHO d. e.arrow_forward
- Which alkene is LEAST reactive in acid-catalyzed hydration? all have equal reactivity А. В. c. D. Е.arrow_forwardDevise a synthesis of each compound from an alkyl halide using any other organic or inorganic reagents. SH b. c. CH;CH,CN d. e. CH,CH,OCOCH3 a.arrow_forward9.54 Draw two different routes to each of the following ethers using a Williamson ether synthesis. Indicate the preferred route (if there is one). a. b. C.arrow_forward
- 16.41 Draw the products of each reaction. а. HNO3 H2SO4 'NO2 HO b. AICI3 CI С. H. Br2 FeBr3arrow_forwardIdentify the reagents from a to d.arrow_forwardWhich carbonyl groups in the anticancer drug taxol (Section 5.5) will undergo nucleophilic addition and which will undergo nucleophilic substitution?arrow_forward
