
Concept explainers
Interpretation: Whether carbocation that is farthest to have an octet in below figure is also least stable and has highest potential energy or not should be determined.
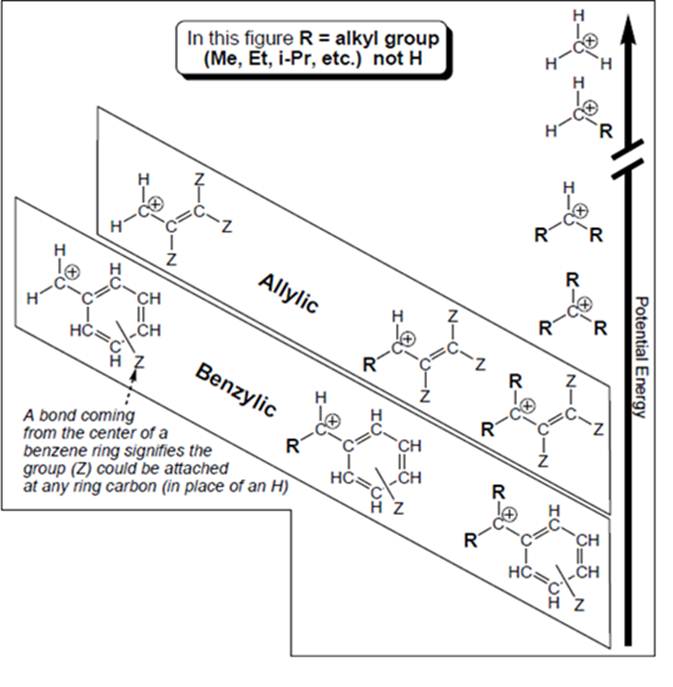
Concept introduction: Organic compounds are covalent in nature that undergoes a reaction by heterolytic cleavage or homolytic cleavage.
In heterolytic cleavage, shared pair of electrons is taken away by one of the atoms which result in charged species. In homolytic cleavage, shared pair of electrons are equally distributed between two atom that results in free radicals.
Carbocation is a general term employed for a postively charged carbon irrespective of valency of carbon. In carbocation, carbon is bonded to 3 atoms or groups and has only sextet of electrons so it behaves as an electron-deficient species. It is
A carbocation is classified as
Want to see the full answer?
Check out a sample textbook solution
Chapter 8 Solutions
Organic Chemistry: A Guided Inquiry
- Build a model of methylcyclohexane, and use the model to complete the following Newmanprojections of methylcyclohexane in the chair conformation: a. When the methyl group is in an axial or equatorial (circle one) position, the molecule is inits lowest potential energy conformation. b. Label one Newman projection above anti and the other gauche to describe the relationshipbetween the methyl group and C3 of the ring. c. In general, which is a lower PE conformation, anti or gauche? d. Explain how your answer to b and c provide an explanation for why it is more favorable fora large group to be in an equatorial than an axial position.arrow_forwardRank these carbocations from the least stable cation to most.arrow_forwardGive a clear handwritten answer..complete the following reaction..choose the perfect arrow for given reactionarrow_forward
- Org. Chem. 1. EA26. Can you please draw out all four , and explain what a stereoisomer is in relation ? Thanksarrow_forwardHow many allylic hydrogens (total number - not type!) are present in the following molecule?arrow_forwardPlease answer this NEATLY, COMPLETELY, and CORRECTLY for an UPVOTE. The stereochemistry of the following alkenes are?arrow_forward
- Arrange the following structures based on increasing reactivity to electrophilic aromatic substitution. CAR OCH₂CH₂CH₂ TAR choose your answer... BAR INCREASING REACTIVITY: choose your answer... COOH JAR choose your answer... V K choose your answer.... Varrow_forwardGive a clear handwritten answer with textual explanation .please give clear handwritten answer...complete the following reaction..arrow_forwardThis is not graded. Please, answer no. 22 & no. 23. Thank you.arrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
