
Organic Chemistry - Standalone book
10th Edition
ISBN: 9780073511214
Author: Francis A Carey Dr., Robert M. Giuliano
Publisher: McGraw-Hill Education
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 7.7, Problem 15P
Place a double bond in the carbon skeleton shown so as to represent
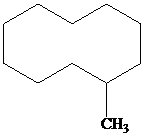
a)
b)
c)
d)
e)
f)
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
The ACS exam at the end of this semester covers a limited amount of IUPAC
nomenclature mostly from Organic I. An example is given below, what is the IUPAC
name of of the compound whose structure is shown in the box?
A) E-1- butylidene-1-iodo-propane
B) E-5-iodo-4-heptene
C) E-1-(1-iodopropylidene)-butane
D) E-3-iodo-3-heptene
A
В
6. What is the correct IUPÁC name of the following compound?
A) (2S, 3S)-3-bromo-2-butanol
B) (2R, 3S)-2-bromo-3-butanol
C) (2R, 3S)-3-bromo-2-butanol
D) (2R, 3R)-2-bromo-3-butanol
E) (2R, 3R)-3-bromo-2-butanol
F) (2S, 3R)-2-bromo-3-butanol
G) (2S, 3R)-3-bromo-2-butanol
H) (2S, 3S)-2-bromo-3-butanol
CH3
HFOH
-Br
The IUPAC name of the compound
is
(a) 5-formylhex-2-en-3-one
(b) 5-methyl-4-oxohex-2-en-5-al
(c) 3-keto-2-methylhex-5-enal
(d) 3-keto-2-methylhex-4-enal
The correct statement regarding electrophile is
(a) electrophile is a negatively charged species
and can form a bond by accepting a pair of
electrons from another electrophile
(b) electrophiles are generally neutral species
and can form a bond by accepting a pair of
electrons from a nucleophile
(c) electrophile can be either neutral or
positively charged species and can form a
bond by accepting a pair of electrons from
a nucleophile
(d) electrophile is a negatively charged species
and can form a bond by accepting a pair of
electrons from a nucleophile,
Which among the given molecules can exhibit
tautomerism?
Ph
Ph
I
III
(a) III only
(b) Both I and III
(d) Both II and III
(c) Both I and II
5)
Which of the following biphenyls is optically
active?
II
Chapter 7 Solutions
Organic Chemistry - Standalone book
Ch. 7.1 - Name each of the following using IUPAC...Ch. 7.1 - Prob. 2PCh. 7.2 - How many carbon atoms are sp2-hybridized in the...Ch. 7.3 - Prob. 4PCh. 7.3 - Are cis-2-hexene and trans-3-hexene stereoisomers?...Ch. 7.4 - Prob. 6PCh. 7.4 - Prob. 7PCh. 7.4 - Give the IUPAC name of each of the compounds in...Ch. 7.5 - Arrange the following in order of increasing...Ch. 7.6 - Prob. 10P
Ch. 7.6 - Standard enthalpies of formation are known for all...Ch. 7.6 - Prob. 12PCh. 7.6 - Despite numerous attempts, the alkene...Ch. 7.6 - Write structural formulas for the six isomeric...Ch. 7.7 - Place a double bond in the carbon skeleton shown...Ch. 7.9 - Identify the alkene obtained on dehydration of...Ch. 7.10 - Prob. 17PCh. 7.11 - Prob. 18PCh. 7.12 - Prob. 19PCh. 7.13 - The alkene mixture obtained on dehydration of...Ch. 7.14 - Write the structures of all the alkenes that can...Ch. 7.14 - Write structural formulas for all the alkenes that...Ch. 7.15 - A study of the hydrolysis behavior of...Ch. 7.15 - Use curved arrows to illustrate the electron flow...Ch. 7.15 - Predict the major product of the reaction shown.Ch. 7.16 - Prob. 26PCh. 7.17 - Prob. 27PCh. 7.18 - Prob. 28PCh. 7.19 - Predict the major organic product of each of the...Ch. 7.19 - A standard method for the synthesis of ethers is...Ch. 7 - Write structural formulas for each of the...Ch. 7 - Prob. 32PCh. 7 - Give an IUPAC name for each of the following...Ch. 7 - A hydrocarbon isolated from fish oil and from...Ch. 7 - Prob. 35PCh. 7 - Prob. 36PCh. 7 - Prob. 37PCh. 7 - Prob. 38PCh. 7 - Choose the more stable alkene in each of the...Ch. 7 - Suggest an explanation for the fact that...Ch. 7 - Prob. 41PCh. 7 - Write structural formulas for all the alkene...Ch. 7 - Prob. 43PCh. 7 - Prob. 44PCh. 7 - Predict the major organic product of each of the...Ch. 7 - Prob. 46PCh. 7 - Prob. 47PCh. 7 - The rate of the reaction In the first order in...Ch. 7 - Prob. 49PCh. 7 - Prob. 50PCh. 7 - You have available 2,2-dimethylcyclopentanol (A)...Ch. 7 - Prob. 52PCh. 7 - Prob. 53PCh. 7 - Prob. 54PCh. 7 - Acid-catalyzed dehydration of...Ch. 7 - The ratio of elimination to substitution is...Ch. 7 - Prob. 57PCh. 7 - Prob. 58DSPCh. 7 - Prob. 59DSPCh. 7 - Prob. 60DSPCh. 7 - Prob. 61DSPCh. 7 - A Mechanistic Preview of Addition Reactions The...Ch. 7 - Prob. 63DSPCh. 7 - Prob. 64DSPCh. 7 - Prob. 65DSP
Additional Science Textbook Solutions
Find more solutions based on key concepts
Fully developed conditions are known to exist for water flowing through a 25-nim-diameer tube at 0.01 kg/s and ...
Fundamentals of Heat and Mass Transfer
Practice Problem ATTEMPT
Write the rate expressions for each of the following reactions:
(a)
(b)
(c)
Chemistry
Write the electron configurations far each of the following elements: (a) Sc. (b) Ti. (c) Cr. (d) Fe. (e) Ru
Chemistry by OpenStax (2015-05-04)
16.43 The following pictures represent solutions at various stages in thetitration of a weak diprotic acid with...
Chemistry (7th Edition)
141. Design a device that uses as electrochemical cell to determine amount of
in a sample water Describe, in...
Chemistry: Structure and Properties (2nd Edition)
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- The the following using IUPAC rules. Select the correct answer from the list below. Z O(Z)-3-methyl-4-propylhept-3-ene (E)-4-ethyl-3-methylhept-3-ene (E)-3-methyl-4-propylhept-3-ene (Z)-4-ethyl-3-methylhept-3-enearrow_forwardWhat is the proper IUPAC name for the compound whose structure is shown? Br A) (R)-3-bromohexane B) (S)-3-bromohexane C) (R)-4-bromohexane D) (S)-4-bromohexane E) None of thesearrow_forwardB The the following using IUPAC rules. Select the correct answer from the list below. Br (Z)-3-bromohex-2-ene (E)-4-bromohex-2-ene (E)-3-bromohex-2-ene O(Z)-4-bromohex-2-enearrow_forward
- H₂N- -Br H₂C Provide the locant and substituent in the order that they will be written in the name. The parent name is (old IUPAC version) or (new IUPAC version).arrow_forwardThe the following using IUPAC rules. Select the correct answer from the list below. Br (E)-4-bromohex-2-ene (Z)-4-bromohex-2-ene (Z)-3-bromohex-2-ene (E)-3-bromohex-2-enearrow_forwarda) Draw the major product(s) of the following reactions. i) ii) OSO4 NaHSO4, H₂O BH3-THF H₂O₂, NaOH b) Methylcyclohexene reacts with chlorine both in CCl4 and water. i) Draw the structural formula for both the products. > ( )arrow_forward
- The IUPAC name of the compound H- is (a) 5-formylhex-2-en-3-one (b) 5-methyl-4-oxohex-2-en-5-al (c) 3-keto-2-methylhex-5-enal (d) 3-keto-2-methylhex-4-enal ( The correct statement regarding electrophile is (a) electrophile is a negatively charged species and can form a bond by accepting a pair of electrons from another electrophile (b) electrophiles are generally neutral species and can form a bond by accepting a pair of electrons from a nucleophile (c) electrophile can be either neutral or ar positively charged species and can form a bond by accepting a pair of electrons from a nucleophile (d) electrophile is a negatively charged species and can form a bond by accepting a pair of electrons from a nucleophile.. Which among the given molecules can exhibit tautomerism? Ph Ph I II III (a) III only (c) Both I and II (b) Both I and III (d) Both II and III 1) Which of the following biphenyls is optically active?arrow_forwardProvide the structures for each compound name given below, including notation for stereochemistry (i.e., R, S, E, Z, etc.) if necessary. a) N-ethylheptanamide b) methyl ethanoate c) 2,8-dimethyl-5-nonanonearrow_forwardIdentify the correct IUPAC name for the following structure.A) 6-bromo-1-cyclopentyl-3,6-dimethylhexaneB) 1-bromo-5-cyclopentyl-1,4-dimethylpentaneC) 2-bromo-6-cyclopentyl-5-methylhexaneD) 5-bromo-1-cyclopentyl-2-methylhexaneE) none of the abovearrow_forward
- Draw the structure of the given compound. Identify if the given name is correct of incorrect. If not, provide the correct IUPAC name of the compound. Don’t use CIS/TRANS or E/Z in naming the compounds. 1) 6-PROPYLCYCOHEPTENE 2) 7-BUTYNYLDODEC-1-ENE Note: need answers immediately. Will give a good rate right away as well.arrow_forward11) Name the IUPAC name of this compound? A) (R)(E)-2-phenyl-4-hexene B) (S)-(Z)-5-phenyl-2-hexene C) (R)-(Z)-2-phenyl-4-hexene D) (3)-(E)-5-phenyl-2-hexenearrow_forward-What of the following statements is false about cyclopentadienyl anion? A) It is aromatic B) It has 5 p molecular orbitals C) It has three full p bonding molecular orbitals D) It has one empty p anti-bonding molecular orbital E) It has two empty p anti-bonding molecular orbital 6. What is the correct IUPAC name of the following compound? A) 1-bromo-3-chloro-5-isobutyl-4-methylbenzene B) 5-bromo-1-chloro-3-sec-propyl-2-methylbenzene C) 5-bromo-1-sec-butyl-3-chloro-2-methylbenzene Br D) 1-bromo-3-sec-butyl-5-chloro-4-methylbenzene E) 5-bromo-1-chloro-3-isobutyl-2-methylbenzene II. Draw the structure for the major organic product of each of the following reaction: 7. CN + -> NC Show strerochemistry if necessary HBr 8. (1 equivalent) (45°C) Show strerochemistnuarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY
Nomenclature: Crash Course Chemistry #44; Author: CrashCourse;https://www.youtube.com/watch?v=U7wavimfNFE;License: Standard YouTube License, CC-BY