
Concept explainers
(a)
Interpretation:
The energy difference between the two chair conformations of
Concept introduction:
The conformation of cyclohexane is a type of
Answer to Problem 7.14P
The energy difference between the two chair conformations of
Explanation of Solution
The two chair conformations of
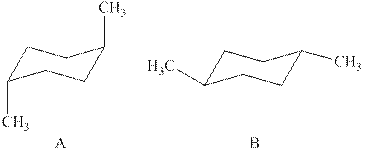
Figure 1
The
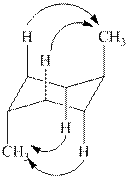
Figure 2
Thus, there are total four
The given value of contribution one
In case of chair conformation B, there is no
Therefore, the energy difference of between the two chair conformations of
Substitute the values of energies in above equation.
Thus, the energy difference is
The energy difference between the two chair conformations of the given compound is
(b)
Interpretation:
The energy difference between
Concept introduction:
The conformation of cyclohexane is a type of
Answer to Problem 7.14P
The energy difference between
Explanation of Solution
The chair conformations of
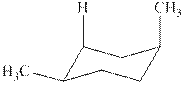
Figure 3
The
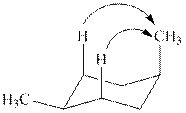
Figure 4
The given value of contribution one
The most stable chair conformation of
![]()
Figure 5
In case of most stable chair conformation of
Therefore, the energy difference of between the two chair conformations of
Substitute the values of energies in above equation.
Thus, the energy difference is
The energy difference between
Want to see more full solutions like this?
Chapter 7 Solutions
Organic Chemistry Study Guide and Solutions
- 3) Draw the most stable conformation of (2Z, 4E)-2,4-hexadiene.arrow_forwardBelow are isomers of tert-butylcyclohexanol in conformational structures. Two are cis-2-tert- butylcyclohexanol and two are cis-3-tert-butylcyclohexanol. Indicate which is the more stable conformation for each pair, and indicate which of all four conformations is the most stable. A OH Ex HO B D OH OHarrow_forwardMethyl isocyanate, CH3 -N= C = O, is used in the industrial synthesis of a type of pesticide and herbicide known as a carbamate. As a historical note, an industrial accident in Bhopal, India, in 1984 resulted in leakage of an unknown quantity of this chemical into the air. An estimated 200,000 people were exposed to its vapors, and over 2000 of these people died. Q.) Methyl isocyanate reacts with strong acids, such as sulfuric acid, to form a cation. Will this molecule undergo protonation more readily on its oxygen or nitrogen atom? In considering contributing structures to each hybrid, do not consider structures in which more than one atom has an incomplete octetarrow_forward
- Draw Newman's projections along the C3¬C4 bond to show the most stable and least stable conformations of 3-ethyl-2,4,4-trimethylheptane.arrow_forwardDraw the structure of an alkane with molecular formula C7H16 that contains (a) one 4° carbon; (b) only 1° and 2° carbons; (c) 1°, 2°, and 3° hydrogens.arrow_forwardwhat is the most stable conformation of (1R, 2S, 5R)-1,2,5-trimethylcyclohexanearrow_forward
- Draw and name the seven aldehydes and ketones with the formula C5H10O. Which are chiral?arrow_forwardDraw the most stable conformation of 1,4-dichlorobutane, using wedges and dashes to represent bonds coming out of the paper and going behind the paper, respectively.arrow_forwardWhich of the following compound is/are meso: * OH CH CI CH CH, CH CH CH OH CI сH CH CH CH CH, II IV I and IV O Il and II O Il and IVarrow_forward
- Are the C¬C bonds cyclopropane weaker than those in cyclohexane?arrow_forward2,3-Dibromoprop-1-ene (C3H4Br2) has four H atoms. Suppose that any of these H atoms can be replaced by a Cl atom to yield a molecule with the formula C3H3Br,Cl. (a) Identify two H atoms where this substitution would yield constitutional isomers of C3H3Br,CI; (b) enantiomers of C3H3Br,Cl; (c) diastereomers of C3H3Br,Cl. H нн H Br Br 2,3-Dibromoprop-1-enearrow_forward5-33 Draw chiral molecules that meet the following descriptions: (a) A chloroalkane, C5H11CI (b) An alcohol, C6H140 (c) An alkene, C6H12 (d) An alkane, C3H18arrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

