
(a)
Interpretation:
The products of the following reactions should be determined and if the products can exist as stereoisomers, it must be shown.
Concept Introduction:
Halogen addition to an alkene:
The addition reaction of halides to
Mainly
In general, the reaction can be shown as below.
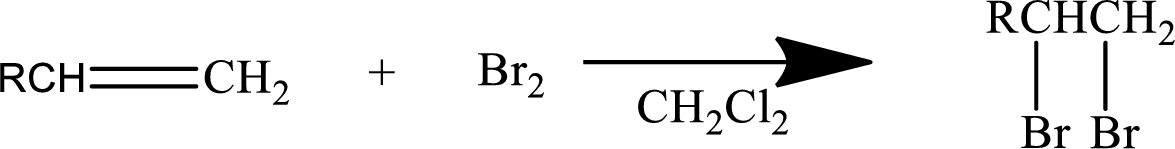
When the bromine approaches the alkene, one bromine accepts the electrons from alkene and give them to the other bromine and a cyclic bromonium ion intermediate is formed.
The formed unstable intermediate reacts with
(b)
Interpretation:
The products of the following reactions should be determined and if the products can exist as stereoisomers, it must be shown.
Concept Introduction:
Acid Catalysed addition of water: When water is added to alkene in the presence of an acid, the product formed will be an alcohol.
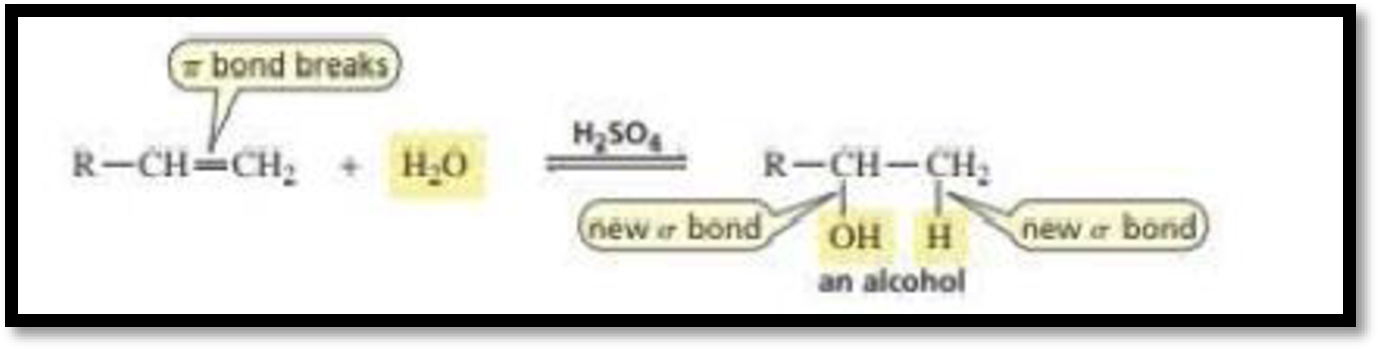
The electrophile
(c)
Interpretation:
The products of the following reactions should be determined and if the products can exist as stereoisomers, it must be shown.
Concept Introduction:
Halogen addition to an alkene:
The addition reaction of halides to alkenes leads to the formation of vicinal dihalide as the product. The two halogens will be on the adjacent carbons.
Mainly
In general, the reaction can be shown as below.
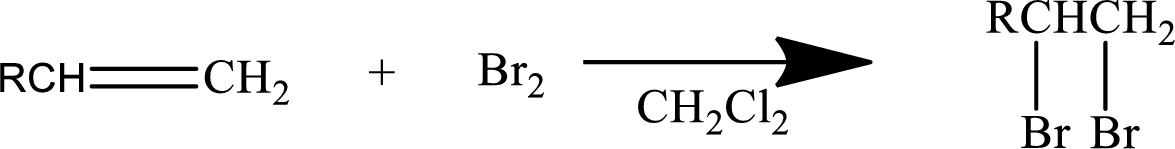
When the bromine approaches the alkene, one bromine accepts the electrons from alkene and give them to the other bromine and a cyclic bromonium ion intermediate is formed.
The formed unstable intermediate reacts with
(d)
Interpretation:
The products of the following reactions should be determined and if the products can exist as stereoisomers, it must be shown.
Concept Introduction:
Addition of a hydrogen halide to an alkene:
An
The
The general reaction is represented as:
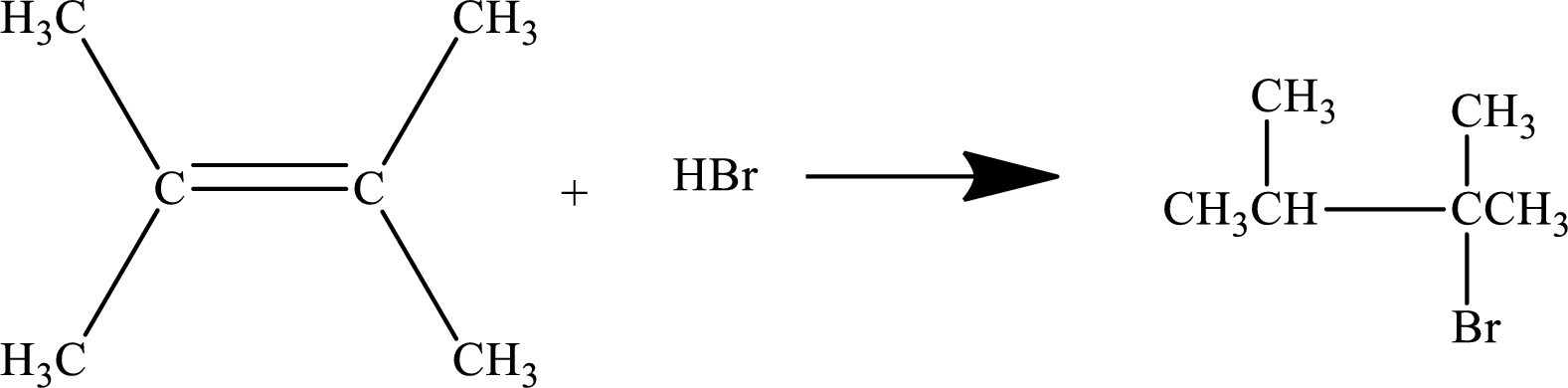
Want to see the full answer?
Check out a sample textbook solution
Chapter 6 Solutions
Organic Chemistry, Books a la Carte Edition (8th Edition)
- Synthesize the products by drawing out reagents and intermediates along the way.arrow_forwardDraw a structural formula for the major product of the reaction shown. -CH₂CH3 Br₂ H₂O • Show product stereochemistry IF the reactant alkene has both carbons of the double bond within a ring. • Do not show stereochemistry in other cases. • If the reaction produces a racemic mixture, just draw one stereoisomer.arrow_forwardDraw the products (including the stereochemistry) formed in the following reaction.arrow_forward
- Predict the products of the following reactions and identify the reactants. Draw the products/reactant. Show the stereochemistry when applicable.arrow_forwardComplete the following reactions (draw the correct products)?arrow_forwardDraw the organic product of the following reaction. CH3 m-CICgH,CO;H • Use the wedge/hash bond tools to indicate stereochemistry where it exists. • If the reaction produces a racemic mixture, just draw one stereoisomer. • If more than one product is possible, only draw the major product. opy astearrow_forward
- Draw the alkene that would react with the reagent given to account for the product formed. ? + HCI My 3 You do not have to consider stereochemistry. • You do not have to explicitly draw H atoms. • In cases where there is more than one answer, just draw one. CH3 CH₂ CHOCH3 TT CI CH3 L ▼ {n [F ? ChemDoodleⓇarrow_forwardDraw the products formed (including stereoisomers) in each reaction.arrow_forwardFill in the missing information (reactants, products, or reagents) in the following transformations. If more than one product is possible, draw structures for all products and indicate the major product. If stereoisomers are possible, draw all isomers, and indicate the major stereoisomers.arrow_forward
- What are the products of the following reactions? Show all stereoisomers that are formed.arrow_forwardIn the presence of heat or light, diazomethane is converted into a carbene that adds to alkenes. Draw the correct products for the following reaction.arrow_forwardSynthesize the product shown from the starting materials, including intermediates formed at each steparrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





