
Concept explainers
The amino acid
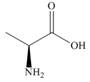
(S)-alanine
a. What is the melting point of
b. How does the melting point of a racemic mixture of
c. What is the specific rotation of
d. What is the optical rotation of a racemic mixture of
e. Label each of the following as optically active or inactive: a solution of pure
(a)
Interpretation: The melting point of
Concept introduction: Enantiomers are the stereoisomers that have exactly opposite
Answer to Problem 5.28P
The melting point of
Explanation of Solution
The given melting point of
The melting point of
(b)
Interpretation: The melting point of a racemic mixture of
Concept introduction: Enantiomers are the stereoisomers that have exactly opposite
Answer to Problem 5.28P
The melting point of a racemic mixture of
Explanation of Solution
The given melting point of
Therefore, the melting point of a racemic mixture of
The melting point of a racemic mixture of
(c)
Interpretation: The specific rotation of
Concept introduction: Enantiomers are the stereoisomers that have exactly opposite
Answer to Problem 5.28P
The specific rotation of
Explanation of Solution
The specific rotation of
Thus, the specific rotation of
The specific rotation of
(d)
Interpretation: The optical rotation of a racemic mixture of
Concept introduction: Enantiomers are the stereoisomers that have exactly opposite
Answer to Problem 5.28P
The optical rotation of a racemic mixture of
Explanation of Solution
The specific rotation of
The specific rotation of
The
Thus, the optical rotation of a racemic mixture of
The optical rotation of a racemic mixture of
(e)
Interpretation: The solutions of pure
Concept introduction: Enantiomers are the stereoisomers that have exactly opposite
Answer to Problem 5.28P
The pure
Explanation of Solution
The pure
A racemic mixture is obtained when
A
Thus, the pure
The pure
Want to see more full solutions like this?
Chapter 5 Solutions
Organic Chemistry
Additional Science Textbook Solutions
General Chemistry: Atoms First
Chemistry: Atoms First
Elementary Principles of Chemical Processes, Binder Ready Version
Organic Chemistry As a Second Language: Second Semester Topics
Chemistry
EBK INTRODUCTION TO CHEMISTRY
- C. Trehalose and isomaltose are both dimers of glucose. However, they have considerably different reactivities. Concisely explain why these differences are observed. OH HO но но. HO HO OH Но- он но OH HO НО trehalose isomaltose 1. Isomaltose is a reducing sugar while trehalose is not. 2. Trehalose is very resistant to acid hydrolysis while isomaltose can be acid-hydrolyzed with ease.arrow_forwardDichlospiroheptane [(a) does (b) does not] have a plane of symmetry; it is [(a) chiral (b) not chiral] A. a,a B. b,b C. a,b D. b,aarrow_forwardExplain the solubility behavior of each compound in water with relevance to their STRUCTURE. c. Amino Acids and Proteins in Water-Alanine (clear colorless solution)-Glutamic acid (clear colorless solution)-Arginine (clear colorless solution)-Albumin (clear colorless solution)-Casein (clear colorless solution)arrow_forward
- 3. Classify the following as a lipid, carbohydrate, or amino acid: (EOCQ 58) CH,OC(CH,)12CH, CHOC(CH,),,CH, CH,0C(CH,),CH=CH(CH,),CH, a.arrow_forwardWhich statement about saccharides is correct? (A). Alpha anomers are more common than beta anoomers. (B). Two monosaccharides can form a disaccharide and oxygen. (C). Polysaccharides with alpha or beta glycosidic linkages have the different properties. (D). Disaccharides can be cyclic or acyclic.arrow_forwardA. Give your comment(s) on the following questions below: 1. Are all chiral molecules enantiomers? Why or why not? 2. What is the difference between enantiomers and diastiomers ? 3. What are the differences between stereoisomers, structural isomers and constitutional isomers?arrow_forward
- 1) When synthesising potential peptide drugs, it is often necessary to use unnatural amino acids. It is essential that we know the stereochemistry of these unnatural amino acids. Below are two line structures of two unnatural amino acids. Draw the S--enantiomer of each of these structures. + H₂N COO CH₂Br مشرق N.arrow_forward36. Indicate the following chemical compound. но OH CH,OH L flavonoid II. aloe-emodin I. aloin IV. coumarine V. taken from Sorreis VI. tannins VII. aglycone of aloin glycoside VIII. taken from Aloe IX. oxymethylantraquinone X. glycoside A) I; I; V; X B) IV; VI; VII C) ; V; VI IX D) II; VII; IX: X E) II; VIII; IX; Xarrow_forward4. Identify the isomers below as structural, cis-trans, or enantiomers. 5.Is this molecule a ketose or aldose sugar?arrow_forward
- O2CCH,CH,-C-CO2 + CH2 NH3 NH3 O2CCH,CH,-C-CO2 + CH2 CH2-C-CO2 + H. phenylalanine -CO2 0,CCH,CH2—CO2 CH2-C-CO2arrow_forwarde Quiz: HW 1: Ch 3: Alkanes AA Quiz: HW 2: Ch 4: Study of Reacti x A Quiz: HW 3: Ch 5: Stereochem A online.butlercc.edu/courses/1479546/quizzes/2495661/take Question 2 If (S)-glyceraldehyde has a specific rotation of -8.7°, what is the specific rotation of (R)- glyceraldehyde? cannot be determined from the information given 0.0° +8.7° -8.7°arrow_forwardMark all chiral center(s) with an asterisk.b. Determine the max number of possible stereoisomers that could be drawn from thisstructure. please explain each step.arrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
 Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning





