
Concept explainers
Interpretation:
The element that is located in the
Concept Introduction:
Periodic law states that if the elements are arranged in increasing order of
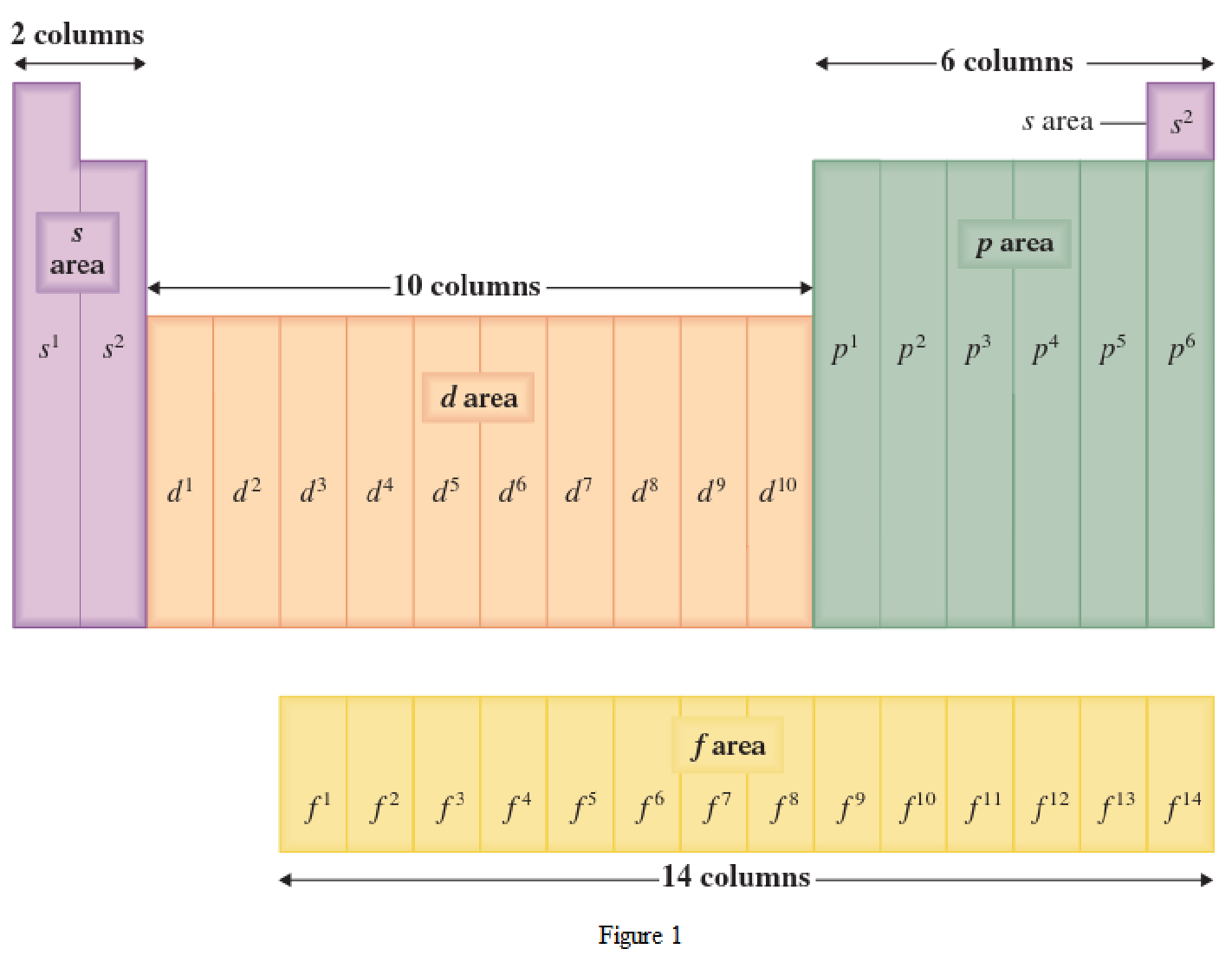
Location of an element in a periodic table can be given by the period number and the group number. The horizontal row in a periodic table where the elements are present is known as Period. The vertical column in a periodic table where the elements are present is known as Group.
Chemical properties of the elements repeat themselves at regular intervals because of the electronic configuration. The elements that are present in a Group have similar chemical properties. This is because the outer-shell electronic configuration will be the same.
The periodic table has all the elements that can be distinguished based on the outer-shell electron. If the outer-shell electron is present in s subshell, then the elements are present in s area of periodic table. If the outer-shell electron is present in p subshell, then the elements are present in p area of periodic table. If the outer-shell electron is present in d subshell, then the elements are present in d area of periodic table. If the outer-shell electron is present in f subshell, then the elements are present in f area of periodic table.
Want to see the full answer?
Check out a sample textbook solution
Chapter 3 Solutions
General, Organic, and Biological Chemistry
- How many unpaired electrons are presents in the orbital diagram for an atom whose electron configuration is1s22s22p4? a. none b. two c. four d. no correct responsearrow_forwardHow many electrons are present in an atom of P1531? a. 15 b. 16 c. 31 d. no correct responsearrow_forwardPart A - Determining the Number of Subatomic Particles in Atoms Which of these atoms has the largest number of neutrons in the nucleus? Check all that apply. 162 Ho 148 Eu O 159 Gd 57 Dy O 149 Nd Submit Request Answer Provide Feedbackarrow_forward
- Test = X Question 36 Order the following ( Lit, 02. N³) according to increasing ionic radius A N3 <02arrow_forward16O and 18O are isotopes of oxygen used by climate scientist as proxies of earth's past temperature. Which of the following is true about these isotopes? a. They contain the same number of neutrons b. They contain the same number of protons c. They contain the same number of electrons d. More than once correct responsearrow_forwardExercise 4.3 Give the number of protons, neutrons, and electrons in the atom symbolized by80201Hgarrow_forwardEXERCISE ON ISOTOPE Isotopic Symbol 6530Zn 32 137BA2* 16S2 56 Atomic No. 30 (4) (8) Nuclear (1) (5) +56 Charge Mass No. 65 (6) 137 No. of (2) (9) protons 16 No. of (3) 16 81 neutrons No. of electrons 30 (7) (10)arrow_forwardWhich of the following elements is located in the d area of the periodic table? a. 20Ca b. 26Fe c. 33As d. no correct responsearrow_forwardProblem 2-2 What is the mass number of an atom containing: (a) 15 protons, 15 electrons, and 16 neutrons? (b) 86 protons, 86 electrons, and 136 neutrons?arrow_forward2:25 1 Question 20 of 25 Submit Solve the following expression for x. What is (are) the value(s) of the positive root(s)? x² = 12.0 (0.200 – x) A) 0.197 B) 11.8, 0.205 C) 12.2 D) There are no positive rootsarrow_forwardWhat is the wavelength of electromagnetic radiation, which corresponds to a frequency of 3.28 x 1018 s-1? m Submit Answer Try Another Version 6 item attempts remaining 38°F Light rain P Type here to search !!arrow_forwardThe Ka for HCN is 4.9 × 10-10. What is the value of Kb for CN-? A) 4.0 × 10-6 B) 2.0 × 109 C) 4.9 × 10-24 D) 4.9 × 104 E) 2.0 × 10-5arrow_forwardarrow_back_iosSEE MORE QUESTIONSarrow_forward_ios
 General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning



