
(a)
Interpretation:
An explanation regarding the arrangement of the compounds in order of their decreasing
Concept introduction:
The
Answer to Problem 3.47AP
The arrangement of the order of decreasing
Explanation of Solution
The given compounds are shown below.
Figure 1
Compounds shown in Figure 1 are alcohols that mean acidic proton is bonded to oxygen.
Compound B contains two chlorine atoms which are electron-withdrawing group. Due to
Compound C contains only one chlorine atom which is an electron-withdrawing group Due to
Compound A contains one methyl group which is an electron-donating group. Due to
Therefore, the order of acidity of the compounds is shown below.
Figure 2
The
Therefore, the order of decreasing
Figure 3
The order of decreasing
The arrangement of the order of decreasing
(b)
Interpretation:
An explanation regarding the arrangement of the compounds in order of their decreasing
Concept introduction:
The
Answer to Problem 3.47AP
The arrangement of the order of decreasing
Explanation of Solution
The given compounds are shown below.
Figure 4
According to the elemental effect, the acidity increases as the
Compound A contains only chlorine atom which is an electron-withdrawing group. Due to
Compound C contains
Therefore, the order of acidity of the compounds is shown below.
Figure 5
The
Therefore, the order of decreasing
Figure 6
The order of decreasing
The arrangement of the order of decreasing
(c)
Interpretation:
An explanation regarding the arrangement of the compounds in order of their decreasing
Concept introduction:
The
Answer to Problem 3.47AP
The arrangement of the order of decreasing
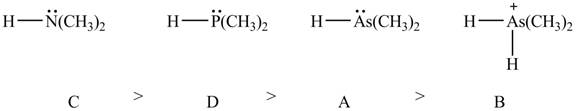
Explanation of Solution
The given compounds are shown below.
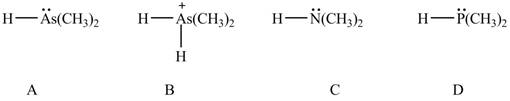
Figure 7
According to the elemental effect, the acidity increases as the atomic number attached to the acidic hydrogen also increases. The element
The atomic number of arsenic is higher than nitrogen and phosphorus. The
The atomic number of phosphorus is higher than nitrogen. The
According to the charge effect, the acidity increases with the presence of positive charge on atom attached to acidic hydrogen. Compound B contains a positive charge. Therefore, compound B is more acidic than compound A.
Therefore, the order of acidity of the compounds is shown below.
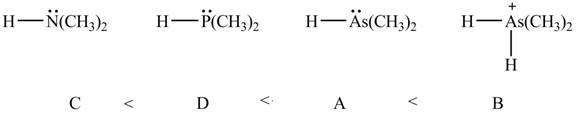
Figure 8
The
Therefore, the order of decreasing
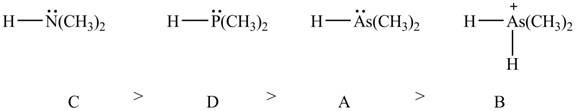
Figure 9
The order of decreasing
The arrangement of the order of decreasing
(d)
Interpretation:
An explanation regarding the arrangement of the compounds in order of their decreasing
Concept introduction:
The
Answer to Problem 3.47AP
The arrangement of the order of decreasing
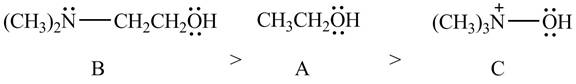
Explanation of Solution
The given compounds are shown below.
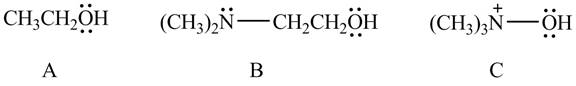
Figure 10
According to the charge effect, the acidity increases with the presence of positive charge on atom attached to acidic hydrogen. Compound C contains a positive charge. Therefore, compound C is more acidic than compound A and B.
Compound B contains only
Therefore, the order of acidity of the compounds is shown below.
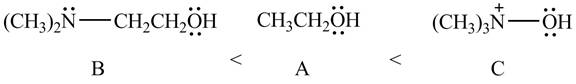
Figure 11
The
Therefore, the order of decreasing
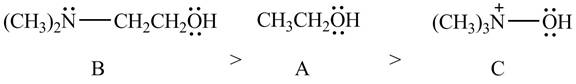
Figure 12
The order of decreasing
The arrangement of the order of decreasing
Want to see more full solutions like this?
Chapter 3 Solutions
Organic Chemistry Study Guide and Solutions
- Will acetylene react with sodium hydride according to the following equation to form a salt and hydrogen, H2? Using pKa values given in Table 4.1, calculate Keq for this equilibrium.arrow_forwardIf the G for a reaction is 4.5 kcal/mol at 298 K, what is the Keq for this reaction? What is the change in entropy of this reaction if H = 3.2 kcal/mol?arrow_forwardIn each equilibrium, label the stronger acid, the stronger base, the weaker acid, and the weaker base. Also estimate the position of each equilibrium. (a) CH3CH2O + CH3CCH CH3CH2OH + CH3CC (b) CH3CH2O + HCl CH3CH2OH + Cl (c) CH3COOH + CH3CH2O CH3COO + CH3CH2OHarrow_forward
- Complete each acid-base reaction and predict whether the position of equilibrium lies toward the left or toward the right. (a) CH3CCH+CH3CH2ONa+CH3CH3OH (b) CH3CCCH2CH2OH+Na+NH2NH3(l)arrow_forward2.) Draw the products of the following reactions. Provide an arrow-pushing mechanism for each reaction. Label the acid, base, conjugate base, and conjugate acid. Finally, indicate the direction of equilibrium. 요요 + NaHarrow_forward5). Which of the following compound has lowest pKa? H3C (A) COOH F (B) COOH 6). What is the product nhtained from the full COOH (C) COOH (D) COOH (E)arrow_forward
- Which of the following compounds are acidic (A), basic (B), or relatively neutral (N). CH;-CH-S-H CH;-CH2-CH3 CH;-CH=CH2 CH;-CH,-0-CH, CH;-CH2-O-Harrow_forwardUsing pKa Values to Determine Relative Acidity and Basicity Rank the following compounds in order of increasing acidity, and then rank their conjugate bases in order of increasing basicity.arrow_forwardF3C - T SOH Br3C III Rank the compounds (shown below) in order of increasing acidity. H3C ОН II ОН A) | < ||| < || B) ||| < || < | C) || < III < | D) ||| < | < || E) II < I < |||arrow_forward
- hydrogens on a carbon adjacent to a carbonyl group are far more acidic than those not adjacent to a carbonyl group. The anion derived from acetone, for example, is more stable than is the anion derived from ethane. Account for the greater stability of the anion from acetone. CH,ČCH, H CH,CH, H Acetone Ethane pK, 20.2 pK, 51arrow_forwardAcetic acid, CH3COOH, is a weak organic acid, pKa 4.76. Write an equation for the equilibrium reaction of acetic acid with each base. Which equilibria lie considerably toward the left? Which lie considerably toward the right? Q.) NaOHarrow_forwardBenzoic acid (C6H5COOH) and aniline (C6H5NH2) areboth derivatives of benzene. Benzoic acid is an acid withKa = 6.3 x10-5 and aniline is a base with Ka = 4.3 x10-10. (a) What are the conjugate base of benzoic acid andthe conjugate acid of aniline? (b) Anilinium chloride(C6H5NH3Cl) is a strong electrolyte that dissociates intoanilinium ions (C6H5NH3+) and chloride ions. Which willbe more acidic, a 0.10 M solution of benzoic acid or a 0.10M solution of anilinium chloride? (c) What is the value ofthe equilibrium constant for the following equilibrium?C6H5COOH(aq) + C6H5NH2(aq) ⇌ C6H5COO-(aq) + C6H5NH3+(aq)arrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
