
Concept explainers
Inhibition of Purine and Pyrimidine
(a)
Interpretation:
A reaction of purine or pyrimidine metabolism is affected by the azaserine.
Concept Introduction:
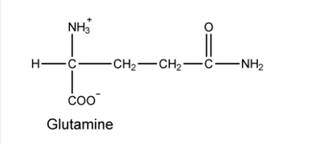
Azaserine and DON are equivalents of glutamine and bind to glutamine-binding proteins. They can react with nucleophiles, leading to covalent modification and inactivation.
Answer to Problem 4P
Azaserine and DON are equivalents of glutamine and bind to glutamine-binding proteins. They can react with nucleophiles, leading to covalent modification and inactivation. The following reactions in purine synthesis are sensitive to these inhibitors.
There is another reaction that's sensitive to those inhibitors for the synthesis of pyrimidines.
Explanation of Solution
There are 3 structures that are analogs to amino acid, and that they are azaserine (O-diazoacetvl-Lserine) and a connected compound DON, and 6-diaxo-5-oxo-L-norleucine. Their structures are shown below:
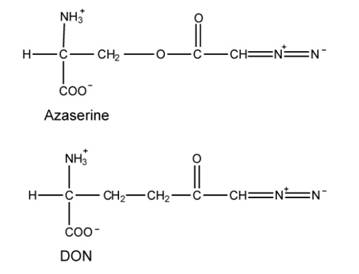
Azaserine and DON are equivalents of glutamine and bind to glutamine-binding proteins. They can react with nucleophiles, leading to covalent modification and inactivation. The following reactions in purine synthesis are sensitive to these inhibitors.
There is another reaction that's sensitive to those inhibitors for the synthesis of pyrimidines.
(b)
Interpretation:
A reaction of purine or pyrimidine metabolism is affected by the methotrexate.
Concept Introduction:
Methotrexate is analogous to dihydrofolate in this it competes with folic acid with high affinity for binding sites on enzymes. Some reactions in purine synthesis are affected.
Answer to Problem 4P
The formation of tTMP in pyrimidine synthesis will be blocked by methotrexate sodium. The reaction is seen below.
Explanation of Solution
Methotrexate is analogous to dihydrofolate in this it competes with folic acid with high affinity for binding sites on enzymes. Some reactions in purine synthesis are affected.
The formation of tTMP in pyrimidine synthesis will be blocked by methotrexate sodium. The reaction is seen below.
(c)
Interpretation:
A reaction of purine or pyrimidine metabolism is affected by the sulfonamides.
Concept Introduction:
Sulfonamides are structurally the same as para-aminobenzoic acid. They inhibit the assembly of folacin in bacterium.
Answer to Problem 4P
Ester synthesis in animals won't be littered with sulfonamides since folacin is an element of a dietary demand for animals.
Explanation of Solution
Sulfonamides are structurally the same as para-aminobenzoic acid. They inhibit the assembly of folacin in bacterium. This inhibition can happen as a result of they cannot produce folacin because of lack of substrate. Ester synthesis in animals won't be littered with sulfonamides since folacin is an element of a dietary demand for animals.
(d)
Interpretation:
A reaction of purine or pyrimidine metabolism is affected by the allupurinol.
Concept Introduction:
Xanthine enzyme can hydroxylate Zyloprim, a suicide substance of organic compound enzyme, to create alloxantine that binds to the catalyst and inactivates it.
Answer to Problem 4P
Organic compound is regenerate to uric acid via the catalyst organic compound enzyme, and excrement acids will not form as a result of its blocked because of each nucleoside and nucleoside that are metabolized to organic compound.
Explanation of Solution
Xanthine enzyme can hydroxylate Zyloprim, a suicide substance of organic compound enzyme, to create alloxantine that binds to the catalyst and inactivates it. Organic compound is regenerate to uric acid via the catalyst organic compound enzyme, and excrement acids will not form as a result of its blocked because of each nucleoside and nucleoside that are metabolized to organic compound.
(e)
Interpretation:
A reaction of purine or pyrimidine metabolism is affected by the 5-fluorouracil.
Concept Introduction:
5-fluorouracil is converted to 5-fluorodeoxyuridylate (FdUMP), which is a powerful inhibitor of thymidylate synthase.
Answer to Problem 4P
Even though 5-fluorouracil is not a significant inhibitor of nucleotide metabolism, it is converted to 5-fluorodeoxyuridylate (FdUMP), which is a powerful inhibitor of thymidylate synthase.
Explanation of Solution
Even though 5-fluorouracil is not a significant inhibitor of nucleotide metabolism, it is converted to 5-fluorodeoxyuridylate (FdUMP), which is a powerful inhibitor of thymidylate synthase.
Want to see more full solutions like this?
Chapter 26 Solutions
Biochemistry
- Diazepam is converted in vivo to an active metabolite Oxazepam. Explain metabolic transfomation of diazepam to oxazepam. (Provide names of enzymes involved in this transformation. Drawings of structures ofintemediate metabolites is NOT required)arrow_forwardGive the pharmacological property of the following terpenes/terpenoids. Where do they naturally occur? saponins terpenoids cardiac glycosides How can terpenes/terpenoids be extracted from plants? What are the chemical tests used in identifying the presence of cardiac glycosides?arrow_forwardWhat is bioavilability of dacarbazine?arrow_forward
- One treatment for hyperuricemia is administration of xanthine oxidase inhibitors like allopurinol. What is the biochemical rationale for this treatment? Discuss the mechanism and show an illustration how this drug able to alleviate symptoms of hyperuricemia?arrow_forwardWhich of the following conversions require more than one step? 1. Alanine Ⓡpyruvate 2. Aspartate Ⓡoxaloacetate 3. Glutamate Ⓡa-ketoglutarate 4. Phenylalanine Ⓡhydroxyphenylpyruvate 5. Proline® glutamate O1 and 4 O 1, 2, and 4 O 1, 3, and 5 O2, 4, and 5 04 and 5 घarrow_forwardExplain the metabolism, biochemical importance and inborn error of Glycine Phenylalanine, tyrosine - Tryptophan Sulphur containing amino acids.arrow_forward
- In 2-page worth of words (around 500), discuss CYCLAMATE's regulation, allowable levels, and what group of people are at high risk for the side effects of CYCLAMATE in the body.arrow_forward6-Mercaptopurine , after its conversion to the corresponding nucleotide through salvage reactions, is a potent competitive inhibitor of IMP in the pathways for AMP and GMP biosynthesis. It is therefore a clinically useful anticancer agent. The chemotherapeutic effectiveness of 6 mercaptopurine is enhanced when it is administered with allopurinol. Explain the mechanism of this enhancement.arrow_forwardWhat is the biochemical basis of allopurinol treatment for gout?arrow_forward
- (a) Fill in the blank regarding the mechanism of chymotrypsin below. (1) Polypeptide substrate binds non covalently in the enzyme active site. The catalytic Triad includes a reactive Nucleophile that attacks the electrophilic amide C atom. (2) The resulting tetrahedral - Is stabilized by H-bonding interactions with the hole.(Note: this is the same word repeated) (3) Collapse of the tetrahedral intermediate and H * Transfer from- Lead to the Cleavage of the C-N bond. The N-terminal peptide is bound through acyl linkage to serine. (4) A Molecule then binds to the active site and attacks the acyl ester car- bonyl. (5) The resulting tetrahedral Tions with the Same word repeated). (6) The second peptide fragment is released, and the enzyme returns to its initial state. Intermediate is stabilized via enthalpic interac- hole. (Note: this is the same answer as (2), and is again the (b) Trypsin and chymotrypsin are two closely related proteases; however, trypsin cleaves after positively charged…arrow_forwardMethyl-labeled [14C]methionine at a specific activity of 2.0 millicuries per millimole was injected into rats. Six hours later the rats were killed. Phosphatidylcholine was isolated from the liver and found to have a specific activity of 1.5 millicuries per millimole. Calculate the proportions of phosphatidylcholine synthesized by the phosphatidylserine pathway and by the pathway starting from free choline. What further information would you need for your calculated values to reflect the true rates of these processes?arrow_forwardPresent the rationale for clinical applications of niacin. What are major side effects?arrow_forward
 BiochemistryBiochemistryISBN:9781319114671Author:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.Publisher:W. H. Freeman
BiochemistryBiochemistryISBN:9781319114671Author:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.Publisher:W. H. Freeman Lehninger Principles of BiochemistryBiochemistryISBN:9781464126116Author:David L. Nelson, Michael M. CoxPublisher:W. H. Freeman
Lehninger Principles of BiochemistryBiochemistryISBN:9781464126116Author:David L. Nelson, Michael M. CoxPublisher:W. H. Freeman Fundamentals of Biochemistry: Life at the Molecul...BiochemistryISBN:9781118918401Author:Donald Voet, Judith G. Voet, Charlotte W. PrattPublisher:WILEY
Fundamentals of Biochemistry: Life at the Molecul...BiochemistryISBN:9781118918401Author:Donald Voet, Judith G. Voet, Charlotte W. PrattPublisher:WILEY BiochemistryBiochemistryISBN:9781305961135Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougalPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305961135Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougalPublisher:Cengage Learning BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning Fundamentals of General, Organic, and Biological ...BiochemistryISBN:9780134015187Author:John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. PetersonPublisher:PEARSON
Fundamentals of General, Organic, and Biological ...BiochemistryISBN:9780134015187Author:John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. PetersonPublisher:PEARSON





