
Organic Chemistry
9th Edition
ISBN: 9781305080485
Author: John E. McMurry
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 24.6, Problem 10P
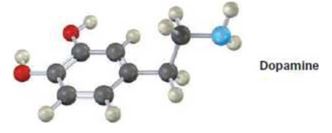
Show two methods for the synthesis of dopamine, a neurotransmitter involved in regulation of the central nervous system. Use any
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
Name the following compound:
O Butyl cyclohexanoate
O Butanoic cyclohexanecarboxylic anhydride
O Cyclohexyl butanoate
O Cyclohexanoic butanoate
O Butyl cyclohexyl anhydride
With reference to the structures of acetylsalicylic acid (aspirin) and acetaminophen (the active ingredient in Tylenol), explain why acetaminophen tablets can be stored in the medicine cabinet for years, but aspirin tablets slowly decompose over time.
What carboxylic acid and amine are needed to synthesize the pain reliever phenacetin? Phenacetin was once a component of the over-the-counter pain reliever APC (aspirin, phenacetin, caffeine), but it is no longer used because of its kidney toxicity.
Chapter 24 Solutions
Organic Chemistry
Ch. 24.1 - Name the following compounds:Ch. 24.1 - Prob. 2PCh. 24.1 - Prob. 3PCh. 24.3 - Prob. 4PCh. 24.3 - The benzylammonium ion (C6H5CH2NH3+) has pKa =...Ch. 24.4 - Prob. 6PCh. 24.5 - Prob. 7PCh. 24.6 - Prob. 8PCh. 24.6 - Prob. 9PCh. 24.6 - Show two methods for the synthesis of dopamine, a...
Ch. 24.6 - Prob. 11PCh. 24.6 - Prob. 12PCh. 24.6 - Prob. 13PCh. 24.7 - Prob. 14PCh. 24.7 - Prob. 15PCh. 24.8 - Prob. 16PCh. 24.8 - Propose syntheses of the following compounds from...Ch. 24.8 - How would you prepare the following compounds from...Ch. 24.8 - Prob. 19PCh. 24.9 - Draw an orbital picture of thiazole. Assume that...Ch. 24.9 - Prob. 21PCh. 24.9 - Prob. 22PCh. 24.9 - Which nitrogen atom in the hallucinogenic indole...Ch. 24.9 - Prob. 24PCh. 24.10 - Prob. 25PCh. 24.SE - Name the following amines, and identify each as...Ch. 24.SE - The following compound contains three nitrogen...Ch. 24.SE - Name the following amine, including R, S...Ch. 24.SE - Prob. 29VCCh. 24.SE - Predict the product(s) for each reaction below and...Ch. 24.SE - Predict the product(s) and provide the complete...Ch. 24.SE - Prob. 32MPCh. 24.SE - Predict the product(s) and provide the mechanism...Ch. 24.SE - The diazotization of aniline first involves the...Ch. 24.SE - Prob. 35MPCh. 24.SE - Prob. 36MPCh. 24.SE - Prob. 37MPCh. 24.SE - Prob. 38MPCh. 24.SE - Choline, a component of the phospholipids in cell...Ch. 24.SE - Prob. 40MPCh. 24.SE - -Amino acids can be prepared by the Strecker...Ch. 24.SE - Prob. 42MPCh. 24.SE - Prob. 43MPCh. 24.SE - Prob. 44MPCh. 24.SE - Propose a mechanism for the following reaction:Ch. 24.SE - Prob. 46MPCh. 24.SE - Name the following compounds:Ch. 24.SE - Prob. 48APCh. 24.SE - Prob. 49APCh. 24.SE - Prob. 50APCh. 24.SE - Histamine, whose release in the body triggers...Ch. 24.SE - Prob. 52APCh. 24.SE - Prob. 53APCh. 24.SE - Prob. 54APCh. 24.SE - Prob. 55APCh. 24.SE - Prob. 56APCh. 24.SE - Prob. 57APCh. 24.SE - Prob. 58APCh. 24.SE - Prob. 59APCh. 24.SE - Prob. 60APCh. 24.SE - Show the products from reaction of p-bromoaniline...Ch. 24.SE - Prob. 62APCh. 24.SE - How would you prepare the following compounds from...Ch. 24.SE - Prob. 64APCh. 24.SE - Phenacetin, a substance formerly used in...Ch. 24.SE - Prob. 66APCh. 24.SE - Draw the structure of the amine that produced the...Ch. 24.SE - Fill in the missing reagents a-e in the following...Ch. 24.SE - Prob. 69APCh. 24.SE - Prob. 70APCh. 24.SE - Deduce the structure of the compound with formula...Ch. 24.SE - Prob. 72APCh. 24.SE - Prob. 73APCh. 24.SE - Prob. 74APCh. 24.SE - Prob. 75APCh. 24.SE - Prob. 76APCh. 24.SE - Propose a structure for the product with formula...Ch. 24.SE - Prob. 78APCh. 24.SE - Prob. 79APCh. 24.SE - Prob. 80APCh. 24.SE - Prob. 81APCh. 24.SE - Prob. 82APCh. 24.SE - Prob. 83APCh. 24.SE - Prob. 84AP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- 18-31 Formic acid is one of the components responsible for the sting of biting ants and is injected under the skin by bee and wasp stings. The pain can be relieved by rubbing the area of the sting with a paste of baking soda (NaHCO3) and water, which neutralizes the acid. Write an equation for this reaction.arrow_forwardWhat two functional groups react to form the following? a. A hemiacetal b. An acetal c. A ketal d. A hemiketalarrow_forwardRank the labeled nitrogen atoms in each compound in order of increasing basicity. Histamine causes the runny nose and watery eyes associated with allergies, and trazodone is a drug used as a sedative and antidepressant.arrow_forward
- Which combination of starting compounds should be chosen to prepare pentyl propanoate pentanoic acid and propanol pentanol and propanoic acid pentanol and pentanoic acid propanol and propanoic acidarrow_forwardWhat product is formed from the addition of NaOH to p-tertbutylphenol? Group of answer choices a compound with addition of an OH group to p-tertbutyl phenol the sodium salt of p-tertbutylphenol, sodium p-tertbutylphenoxide a protonated version of p-tertbutylphenol there is no reactionarrow_forward18-46 Procaine (its hydrochloride salt is marketed as Novocaine) was one of the first local anesthetics developed for infiltration and regional anesthesia. it is synthesized by the following fischer esterification:arrow_forward
- Draw the structure of the neutral product formed in the following reaction. C,H,0¯ C,H,OHarrow_forwardWhat is the reduction product of the following compound with H2/Pd? A 2-propyl-1-cyclohexanol B 2-propenyl-1-cyclohexanol C2-propylcyclohexanone D 1-propyl-2-cyclohexanol What is the oxidation product of this compound? H' A ethanoic acid acetic acid propanoic acid D propanone What is the oxidation product of this compound? A 3,4-dimethylpentanoic acid B 2,3-dimethylpentanoic acid C2,3-dimethylpentanone 3,4-dimethylpentanonearrow_forwardBisphenol A is widely used as a building block in polymer synthesis and is found in the polycarbonate hard plastics of reusable drink containers, DVDs, cell phones, and other consumer goods. Bisphenol A is reported to have estrogenic activity, and its widespread occurrence in our environment is a potential concern. Describe one or two biochemical experiments that could be done to compare the activity of bisphenol A with that of its estradiol, its structural relative.arrow_forward
- Ethyl butyrate, CH3CH2CH2CO2CH2CH3CH3CH2CH2CO2CH2CH3, is an artificial fruit flavor commonly used in the food industry for such flavors as orange and pineapple. Its fragrance and taste are often associated with fresh orange juice, and thus it is most commonly used as orange flavoring. It can be produced by the reaction of butanoic acid with ethanol in the presence of an acid catalyst (H+H+): CH3CH2CH2CO2H(l)+CH2CH3OH(l)H+⟶CH3CH2CH2CO2CH2CH3(l)+H2O(l)CH3CH2CH2CO2H(l)+CH2CH3OH(l)⟶H+CH3CH2CH2CO2CH2CH3(l)+H2O(l) A chemist ran the reaction and obtained 5.40 g of ethyl butyrate. What was the percent yield, The chemist discovers a more efficient catalyst that can produce ethyl butyrate with a 78.0% yield. How many grams would be produced from 7.45g of butanoic acid and excess ethanol?arrow_forwardWhen trichloroacetaldehyde is dissolved in water, almost all of it is converted to the hydrate. Chloral hydrate, the product of the reaction, is a sedative that can be lethal. A cocktail laced with it is known—in detective novels,at least—as a “Mickey Finn.” Explain why an aqueous solution of trichloroacetaldehyde is almost all hydrate.arrow_forwardCardiolipins are found in heart muscles. Draw the products formed when a cardiolipin undergoes complete acid-catalyzed hydrolysis.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning


Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:9781285869759
Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:Cengage Learning

Chemistry for Today: General, Organic, and Bioche...
Chemistry
ISBN:9781305960060
Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:Cengage Learning
Characteristic Reactions of Benzene and Phenols; Author: Linda Hanson;https://www.youtube.com/watch?v=tjEqEjDd87E;License: Standard YouTube License, CC-BY
An Overview of Aldehydes and Ketones: Crash Course Organic Chemistry #27; Author: Crash Course;https://www.youtube.com/watch?v=-fBPX-4kFlw;License: Standard Youtube License