
General, Organic, and Biological Chemistry - 4th edition
4th Edition
ISBN: 9781259883989
Author: by Janice Smith
Publisher: McGraw-Hill Education
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
Chapter 24, Problem 26P
Interpretation Introduction
(a)
Interpretation:
The reason due to which an isomerase enzyme is used in the conversion of mannose-6-phosphate to fructose-6-phosphate needs to be explained.
Concept Introduction:
- Enzymes are biological catalysts that
speed up the reactions taking place within the cells. - Isomers are the compounds with same molecular formula but different arrangement of atoms.
- Mannose (C6H12O6) is −
- Fructose (C6H12O6) is −
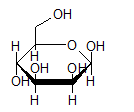
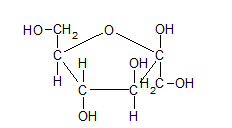
Interpretation Introduction
(b)
Interpretation:
The type of isomers represented by mannose-6-phosphate and fructose-6-phosphate needs to be explained.
Concept Introduction:
- Enzymes are biological catalysts that speed up the reactions taking place within the cells.
- Isomers are the compounds with same molecular formula but different arrangement of atoms.
- Mannose (C6H12O6) is −
- Fructose (C6H12O6) is −
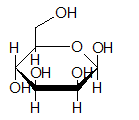
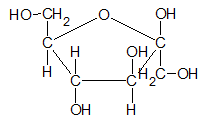
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
(a) Are galactose and mannose constitutional isomers or stereoisomers? (b) Draw the structure of galactose 1-phosphate and mannose 6-phosphate. (c) Are these two phosphates constitutional isomers or stereoisomers?
What is the product of the reaction when α-amylase acts on amylose?
(A) amylose molecules
(B) monosaccharide molecules
(C) different-sized oligosaccharide fragments
(D) different-sized polysaccharide fragments
(i) Which one of the following is a polysaccharide :Starch, Maltose, Fructose, Glucose?(ii) What one difference between a-helix and P-pleated sheet structure of protein.(iii) Write the name of the disease caused by the deficiency of Vitamin B12.
Chapter 24 Solutions
General, Organic, and Biological Chemistry - 4th edition
Ch. 24.2 - Analyze the following reaction by considering the...Ch. 24.2 - Prob. 24.2PPCh. 24.3 - Prob. 24.1PCh. 24.3 - Prob. 24.2PCh. 24.3 - Prob. 24.3PCh. 24.3 - Prob. 24.4PCh. 24.3 - Prob. 24.5PCh. 24.3 - Prob. 24.6PCh. 24.4 - Prob. 24.7PCh. 24.4 - Prob. 24.8P
Ch. 24.4 - Prob. 24.9PCh. 24.5 - Prob. 24.10PCh. 24.5 - Prob. 24.11PCh. 24.5 - Prob. 24.12PCh. 24.6 - Prob. 24.13PCh. 24.7 - Prob. 24.14PCh. 24.7 - Prob. 24.3PPCh. 24.7 - Prob. 24.15PCh. 24.7 - Prob. 24.16PCh. 24.7 - Use the number of molecules of ATP formed from the...Ch. 24.7 - Prob. 24.18PCh. 24.8 - Prob. 24.19PCh. 24.8 - Prob. 24.20PCh. 24.8 - Prob. 24.21PCh. 24.9 - Prob. 24.4PPCh. 24.9 - What products are formed when each amino acid is...Ch. 24.9 - Prob. 24.22PCh. 24 - Analyze each reaction by considering the...Ch. 24 - Analyze each reaction by considering the...Ch. 24 - Prob. 25PCh. 24 - Prob. 26PCh. 24 - Prob. 27PCh. 24 - Prob. 28PCh. 24 - Prob. 29PCh. 24 - Prob. 30PCh. 24 - Prob. 31PCh. 24 - Prob. 32PCh. 24 - Glucose is completely metabolized to six molecules...Ch. 24 - Why is glycolysis described as an anaerobic...Ch. 24 - Write the overall equation with key coenzymes for...Ch. 24 - Prob. 36PCh. 24 - Prob. 37PCh. 24 - Prob. 38PCh. 24 - Consider the aerobic and anaerobic avenues of...Ch. 24 - Prob. 40PCh. 24 - Prob. 41PCh. 24 - Prob. 42PCh. 24 - Prob. 43PCh. 24 - Prob. 44PCh. 24 - Prob. 45PCh. 24 - Prob. 46PCh. 24 - Prob. 47PCh. 24 - Prob. 48PCh. 24 - Prob. 49PCh. 24 - Prob. 50PCh. 24 - Prob. 51PCh. 24 - Prob. 52PCh. 24 - Prob. 53PCh. 24 - Prob. 54PCh. 24 - Prob. 55PCh. 24 - Prob. 56PCh. 24 - Prob. 57PCh. 24 - Prob. 58PCh. 24 - Prob. 59PCh. 24 - How much ATP is generated by the complete...Ch. 24 - Prob. 61PCh. 24 - Fill in the boxes with the number of moles of each...Ch. 24 - Prob. 63PCh. 24 - Prob. 64PCh. 24 - Prob. 65PCh. 24 - Prob. 66PCh. 24 - Prob. 67PCh. 24 - Prob. 68PCh. 24 - Prob. 69PCh. 24 - Prob. 70PCh. 24 - What is the difference between ketogenic and...Ch. 24 - Prob. 72PCh. 24 - Prob. 73PCh. 24 - Draw the structure of the keto acid formed by the...Ch. 24 - Draw the products formed in each transamination...Ch. 24 - Prob. 76PCh. 24 - Prob. 77PCh. 24 - Prob. 78PCh. 24 - Prob. 79PCh. 24 - Prob. 80PCh. 24 - What metabolic intermediate is formed from the...Ch. 24 - What metabolic intermediate is formed from the...Ch. 24 - Prob. 83PCh. 24 - Prob. 84PCh. 24 - Prob. 85PCh. 24 - Prob. 86PCh. 24 - Prob. 87PCh. 24 - What is the cause of the pain and cramping in a...Ch. 24 - Prob. 89PCh. 24 - Prob. 90PCh. 24 - Prob. 91PCh. 24 - Prob. 92PCh. 24 - Prob. 93PCh. 24 - Prob. 94PCh. 24 - What type of enzyme would catalyze the conversion...Ch. 24 - Prob. 96PCh. 24 - Prob. 97CPCh. 24 - Prob. 98CPCh. 24 - Prob. 99CPCh. 24 - Prob. 100CP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- In glycoside formation, the hemiacetal functional group of a cyclic monosaccharide is converted to an acetal group by reaction with a(an)arrow_forwardWrite the products of oxidation of glucose with:(a) Bromine water (b) Nitric acidarrow_forward(i) Which one of the following is a disaccharide : Starch, Maltose, Fructose, Glucose?(ii) What is the difference between fibrous protein and globular protein?(iii) Write the name of vitamin whose deficiency causes bones deformities in children.arrow_forward
- For the following carbohydrates, please indicate the TRUE alternative: (a) The carbohydrate V is reducing and is hydrolyzed by an enzyme of the type β galactosidase (b) Carbohydrate VI is not reducing or mutating. (c) Carbohydrates I and IV have β-glucosidic bond, while carbohydrate II has α-glucosidic bond. (d) Carbohydrate III is neither reducing nor hydrolyzed by an α-type enzyme such as maltase. (e) Carbohydrate I is reducing and mutating.arrow_forward(i) Which one of the following is an oligosaccharide: starch, maltose, fructose, glucose (ii) What is the difference between DNA and RNA. (iii) Write the name of the disease caused by the deficiency of vitamin B1arrow_forwardDoes lactose mutarotate?arrow_forward
- In strong base, glucose converts to fructose. Explain howthis conversion occurs.arrow_forwardCrocin, which occurs naturally in crocus and gardenia flowers, is primarily responsible for the color of saffron. (a) What lipid and monosaccharides are formed by the hydrolysis of crocin? (b) Classify the lipid as a monoterpenoid, diperpenoid, etc., and locate the isoprene units.arrow_forwardIf an enzyme-catalyzed reaction has a high rate at low pH and low rate at higher pH, this implies that a group on either the enzyme or the substrate must be for an efficient reaction. leaving group oxidoreductase coenzymes O protonated deprotonated The compound that consists of deoxyribose linked by an N-glycosidic bond to N-9 of guanine is: adenylate deoxyguanosine guanosine nucleotide guanylatearrow_forward
- Define fermentation. Name the enzyme which converts(a) milk into curd (yogurt)(b) Cane sugar into glucose and fructose(c) glucose into ethanolarrow_forwardIf you take crystals of the disaccharide lactose the natural substrate for beta-galactosidase and drop them in water, why doesn’t the lactose immediately react with the water and come apart into glucose and galactose?arrow_forward1. Among the following five types of biomolecules, which is the least common drug target for commercial drugs? (a) Enzymes (b) Receptors (c) Carbohydrates (d) Membrane transport proteins (e) DNA 2. Which is a false statement regarding non-competitive enzyme inhibitor? (a) It binds to an allosteric site of the enzyme (b) It does not compete with the natural substrate for binding to the active site of the enzyme (c) Its binding to the enzyme can induce a conformational change of the enzyme (d) It can be used in combination therapies with a competitive inhibitor (e) It usually is a mimic of the natural enzyme substrate 3. What is the purpose of structure-activity-relationship (SAR) studies? (a) To identify the drug target (b) To improve the pharmacokinetic properties of the drug (c) To determine which atoms or functional groups are important to the activity of a drug (d) To simplify the synthesis of the drug (e) To optimise activity of the drug 4. Among the following five types of…arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:9781285869759
Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning