
Organic Chemistry
7th Edition
ISBN: 9780321803221
Author: Paula Y. Bruice
Publisher: Prentice Hall
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 23, Problem 45P
a. Explain why the
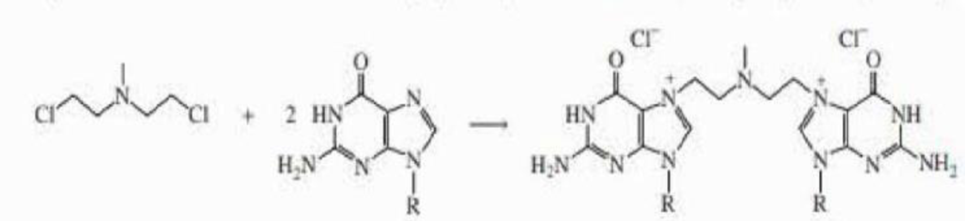
b. The alkyl halide can react with two guanines, each in a different DNA chain, thereby cross-linking the chains. Propose a mechanism for the cross-linking reaction.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
a. Explain why the alkyl halide shown here reacts much more rapidly with guanine than does a primary alkyl halide (such as pentyl chloride).
b. The alkyl halide can react with two guanines, each in a different DNA chain, thereby cross-linking the chains. Propose a mechanism for thecross- linking reaction.
Explain why the alkyl halide shown here reacts much more rapidly with guanine than does a primary alkyl halide (such as pentyl chloride). The alkyl halide can react with two guanines, each in a different DNA chain, thereby cross-linking the chains. Propose a mechanism for the cross-linking reaction.
Draw the product of the Lewis acid-base reaction. Label the electrophile and nucleophile.
Yo
FO
nucleophile
+
BF3
electrophile
F3B-
✓
edit structure ...
Chapter 23 Solutions
Organic Chemistry
Ch. 23.2 - Compare each of the mechanisms listed here with...Ch. 23.2 - Prob. 3PCh. 23.2 - Prob. 4PCh. 23.3 - a. Draw the mechanism for the following reaction...Ch. 23.5 - Prob. 7PCh. 23.5 - Propose a mechanism for the Co2+ catalyzed...Ch. 23.6 - Prob. 9PCh. 23.7 - Prob. 10PCh. 23.7 - Prob. 12PCh. 23.7 - Prob. 13P
Ch. 23.9 - Which of the following amino acid side chains can...Ch. 23.9 - Which of the following C-terminal peptide bonds is...Ch. 23.9 - Carboxypeptidase A has esterase activity as well...Ch. 23.9 - Arginine and lysine side chains fit into trypsins...Ch. 23.9 - Explain why serine proteases do not catalyze...Ch. 23.10 - If H2 18O is used in the hydrolysis reaction...Ch. 23.10 - When apples that have been cut are exposed to...Ch. 23.11 - Prob. 22PCh. 23.11 - The pHactivity profile for glucose-6-phosphate...Ch. 23.11 - Draw the pH-activity profile for an enzyme that...Ch. 23.12 - Prob. 25PCh. 23.12 - Draw the mechanism for the hydroxide ion-catalyzed...Ch. 23.12 - What advantage does the enzyme gain by forming an...Ch. 23.12 - Prob. 28PCh. 23.12 - Aldolase shows no activity if it is incubated with...Ch. 23 - Which of the following parameters would be...Ch. 23 - Prob. 30PCh. 23 - Prob. 31PCh. 23 - Prob. 32PCh. 23 - Indicate the type of catalysis that is occurring...Ch. 23 - The deuterium kinetic isotope effect (KH2O/KD2O)...Ch. 23 - Prob. 35PCh. 23 - Co2+ catalyzes the hydrolysis of the lactam shown...Ch. 23 - there are two kinds of aldolases. Class I...Ch. 23 - Prob. 38PCh. 23 - The hydrolysis of the ester shown here is...Ch. 23 - Prob. 40PCh. 23 - At pH = 12, the rate of hydrolysis of ester A is...Ch. 23 - 2-Acetoxycyclohexyl tosylate reacts with acetate...Ch. 23 - Proof that an imine was formed between aldolase...Ch. 23 - Prob. 44PCh. 23 - a. Explain why the alkyl halide shown here reacts...Ch. 23 - Triosephosphate isomerase (TIM) catalyzes the...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- * Question Completion Status: A Moving to the next question prevents changes to this answer. Question 2 Coupling a terminal alkyne with an alkenyl halide, using a mixture of a Pd and Cu(l) catalyst, is an example of O A. Sonogashira Coupling O B. Suzuki-Miyaura Coupling OC. Heck-Mizoroki Coupling OD. Gilman Reagent-Mediated Coupling O E. Stille Coupling and Carbonylation reaction A Moving to the next question prevents changes to this answer.arrow_forward2. CI The intermediate in the following nucleophilic aromatic substitution reaction has at least 5 resonance structures. For this question, complete the following tasks: a. Show the mechanistic arrow(s) for the first step. b. Draw TWO of the many resonance structures of the intermediate, drawing out the -NO₂ groups in each. c. Draw the neutral product. NO₂ NO₂ HO and others neutral productarrow_forward4. Show the step-by-step process. Do not use shortcut methods. Make it as detailed as it can be. ENCODE (not hand-written)!arrow_forward
- Need help with this SN2 esterification reaction mechanism with pushing arrows. Please help.arrow_forwardPropose a mechanism for the following reaction. a. Explain why two products are formed. b. Explain why methanol substitutes for only one of the brominesarrow_forward4. Show the step-by-step process. Do not use shortcut methods. Make it as detailed as it can be. ENCODE THE ANSWER.arrow_forward
- We have seen that S-adenosylmethionine (SAM) methylates the nitrogen atom of noradrenaline to form adrenaline, a more potent hormone. If SAM methylates an OH group attached to the benzene ring instead, it completely destroys noradrenaline’s activity. a. Show the mechanism for the methylation of the OH group by SAM. b. Which reaction is more apt to occur, methylation on nitrogen or methylation on oxygen?arrow_forwardPlease draw the product for the Morita Baylis Hilman reaction and identify the nucleophile and electrophile in the intermediate step. Show all arrowing pushing steps.arrow_forwarda. Draw the structure of the tetrahedral intermediate INITIALLY-FORMED in the reaction shown. You do not have to consider stereochemistry. Do not include counter-ions, e.g., Na+, I-, in your answer. In cases where there is more than one answer, just draw one b. Draw the structures of the organic products of the acyl transfer reaction. You do not have to consider stereochemistry. Draw the neutral form of the products; no charges. Draw one structure per sketcher. Add additional sketchers using the drop-down menu in the bottom right corner. Separate multiple products using the + sign from the drop-down menu.arrow_forward
arrow_back_ios
arrow_forward_ios
Recommended textbooks for you
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning
Coenzymes and cofactors; Author: CH15 SWAYAM Prabha IIT Madras;https://www.youtube.com/watch?v=bubY2Nm7hVM;License: Standard YouTube License, CC-BY
Aromaticity and Huckel's Rule; Author: Professor Dave Explains;https://www.youtube.com/watch?v=7-BguH4_WBQ;License: Standard Youtube License