
a)
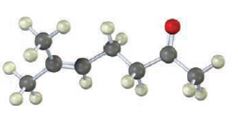
Interpretation:
The steps involved in preparing the compound represented by the model, using either malonic ester synthesis or an acetoacetic ester synthesis, are to be given.
Concept introduction:
Acetoacetic ester synthesis converts an
Both reactions involve the same steps such as i) enolate ion formation ii) SN2 attack of the enolate anion on the alkyl halide iii) hydrolysis and decarboxylation.
Answer to Problem 17VC
The steps involved in preparing the compound represented by the model using acetoacetic ester synthesis are given below.
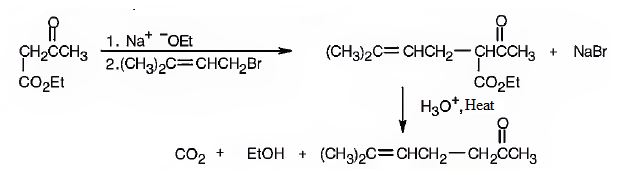
Explanation of Solution
The compound represented by the model is 6-methylhept-5-ene-2-one. It is a methyl ketone and hence it can be prepared using acetoacetic ester synthesis. The ethoxide ion abstracts a proton from the active methylene group of the ester to form the enolate ion. The enolate ion then attacks 1-bromo-3-methy-2-butene and displaces the bromine as bromide ion. The product obtained upon hydrolysis with dilute acids and decarboxylation by heating yields the product.
The steps involved in preparing the compound represented by the model using acetoacetic ester synthesis are given below.

b)
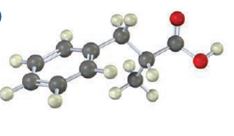
Interpretation:
The steps involved in preparing the compound represented by the model using either malonic ester synthesis or an acetoacetic ester synthesis are to be given.
Concept introduction:
Acetoacetic ester synthesis converts an alkyl halide in to a methyl ketone having three more carbons. The methyl ketone part comes from acetoacetic eater while the remaining carbon comes from the primary alkyl halide. Malonic ester synthesis converts an alkyl halide to a carboxylic acid having two more carbon atoms.
Both reactions involve the same steps such as i) enolate ion formation ii) SN2 attack of the enolate anion on the alkyl halide iii) hydrolysis and decarboxylation.
Answer to Problem 17VC
The steps involved in preparing the compound represented by the model using malonic ester synthesis are given below.
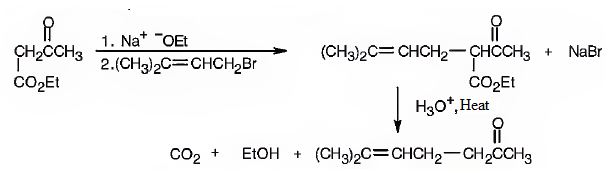
Explanation of Solution
The compound represented by the model is 2-methyl-3-phenylpropanoic acid and hence it can be prepared using malonic ester synthesis. The ethoxide ion abstracts a proton from the active methylene group to form the enolate ion. The enolate ion then attacks benzyl bromide and displaces the bromine as bromide ion. The abstraction of another acidic hydrogen in the product by the base and the nucleophilic displacement of bromine from methyl bromide by enolate ion introduces a methyl group at α- position of the diester. The alkylated diester obtained upon hydrolysis with aqueous acids and decarboxylation by heating yields the product.
The steps involved in preparing the compound represented by the model using malonic ester synthesis are given below.

Want to see more full solutions like this?
Chapter 22 Solutions
EBK ORGANIC CHEMISTRY
- How would you use the acetoacetic ester synthesis to prepare the following? ouarrow_forwardName the carbonyl compound that would be formed by the complete acidic hydrolysis of the following hemiacetal/hemiketal or acetal/ketal: OH OCH₂CH₂CH₂CH₂CH3arrow_forwardPropose a synthesis for each ketone, using as one step in the sequence a Claisen condensation and the reaction sequence illustratedarrow_forward
- Rimantadine is an antiviral drug used to treat people infected with life-threatening influenza viruses. Identify the starting ketone that would be necessary in order to prepare rimantadine via a reductive amination. NH2 Rimantadinearrow_forwardChoose the correct phrase from the following One of the important methods for preparing cyclic amides is the reaction of Crignard reagent with cyclohexanone. The reaction of inulin with indole is given in an acidic medium that gives cyclic amide Caprolactam is a type of cyclic amide that can be prepared from cyclohexane e 2:15arrow_forward1) How might you convert geraniol into either ethyl geranylacetate or geranylacetone? Geraniol CH₂OH CO₂Et Ethyl geranylacetate the Geranylacetonearrow_forward
- Propose a reaction for the formation of the following products involving esterformation.arrow_forwardIndicate how the following compounds can be synthesized from cyclohexanone and any other necessary reagents:arrow_forwardPropose preparative routes to the following ketones using an enamine intermediate - you must make the bond highlighted in red:arrow_forward

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

